Predict the major product from each of the following reactions. H O (1) (2) HO+ (3) HCrO4
Question:
Predict the major product from each of the following reactions.
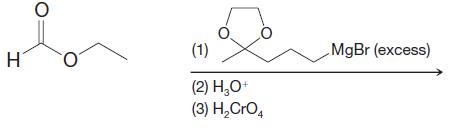
Transcribed Image Text:
H O (1) (2) H₂O+ (3) H₂CrO4 MgBr (excess)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
For the first reaction the major product is expected to be 2butanol This is because the reactant 2br...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict the major organic product from each of the following reaction sequences. (a) (b) (c) (d) (e) (f) (1) MeMgBr (excess) (2) NH,CI, H20 (1) Mg (2) H H (3) HyO (1) PBr3 (2) Mg (3) H,o OH OH (1)...
-
Predict the major organic product from each of the following reactions. (a) (b) (c) (d) (1) CH MgBr (2) HO (1) MgBr (2) NH,CI, H20 MgBr (1 equiv.) O (1) o (2) H,O (1) CH,CHLi (excess) (2) NH,CI, H2O O
-
choess answaer that How many bits are required to store one BCD digit?" 2 8 1 Kilo bits are equal to 1000 bits 1024 bits 1012 bits 1008 bits The function of the encoder is to convert coded...
-
3x > 9 Describe the solution set as an inequality, in interval notation, and on a graph.
-
The following data are the measures of the diameters of 36 rivet heads in 1/100 of an inch.(a) Compute the sample mean and sample standard deviation.(b) Construct a relative frequency histogram of...
-
The stockholders' equity of Elsworth Corporation at January 1 is shown below: The following transactions, among others, occurred during the year: Apr. 1 Declared a 100 percent stock dividend on all...
-
Grass Security, which began operations in 2015, invests in long-term available-for-sale securities. Following is a series of transactions and events determining its long-term investment activity....
-
Hi, Can i get the calculations for Operational Budgets for Plan2go - FNSACC412 How you get the amounts. Thank you
-
The Heinrich Company manufactures two types of plastic hanger racks (Foldaways and Straightaways) especially suited for mounting near clothes dryers. Because permanent press clothing must be hung on...
-
Show how the diketone at the right could be prepared by a condensation reaction: lion to bolo HO R OEt + + R EtOH
-
Provide a mechanism for the following reaction. NO NO 2 ss-sr NaOCH3 OH NO O=5 S=0 NO
-
To help familiarize you with the financial reporting of a real company in order to further your understanding of the chapter material. This case continues our examination of Columbia Sportswear. Here...
-
DLID Co., Ltd, a Hong Kong listed Property Company, has announced a rights offer to raise 3,000,000 new shares issued at a $10.05 subscription price. There are 9,000,000 shares outstanding trading at...
-
The following are transactions for the Sky Blue Corporation: a. Collected $4,650 rent for the period October 1 to December 31, which was credited to Deferred Revenue on October 1. b. Paid $3,000 for...
-
Company ACQ is planning to acquire Company TAR. ACQ currently has 1,800 shares outstanding and a share price of $52. TAR has 1,300 shares outstanding and a share price of $40. The synergy of the...
-
7. (10 points) The diagram at right shows a WIFI network with an access point, X and hosts B and C. The large circles indicate the coverage areas of the three hosts. The coverage area for X is not...
-
Review your ENTJ personality assessment and consider how that has impacted you during Ametros simulation, this course, and the OPM program. Also, project how your personality will influence you as...
-
For Problem 9, suppose the average inflation rate over this period was 3.2 percent and the average T bill rate over the period was 4.3 percent. a. What was the average real return on the stock? b....
-
How does the organizational structure of an MNC influence its strategy implementation?
-
Which of these stereo isomers has the faster rate of E2elimination? D D H Br Br D.
-
Show the products of this elimination reaction and explain which ismajor: t-BUOH PHCHCH CI + 1-BuO
-
Explain why deuterium is lost in preference to hydrogen in this Hofmann eliminationreaction: *N(CH)3 D. H. + (CH)N + HOD
-
Cori's Dog House is considering the installation of a new computerized pressure cooker for hot dogs. The cooker will increase sales by $7,900 per year and will cut annual operating costs by $14,000....
-
Riley Co experiences 20 million loss this year. It is more than likely than not that this Net operating loss will expire unused. How should Riley record this loss as part of calculating its income...
-
( Fixed Income Securities ) 1 8 ) F ) Define the curvature of the term structure with respect to short - term, medium - term and long - term bonds and hence calculate the curvatures of the term...

Study smarter with the SolutionInn App


