Show how the diketone at the right could be prepared by a condensation reaction: lion to bolo
Question:
Show how the diketone at the right could be prepared by a condensation reaction:
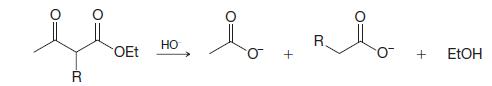
Transcribed Image Text:
lion to bolo HO R OEt + + R EtOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
The d ik et one at the right could be prepared by a cond ensation reaction between two molecules of a lde hyde and two molecules of ket one The reacti...View the full answer

Answered By

DHRUV RAI
As a tutor, I have a strong hands-on experience in providing individualized instruction and support to students of all ages and ability levels. I have worked with students in both one-on-one and group settings, and I am skilled in creating engaging and effective lesson plans that meet the unique needs of each student.
I am proficient in using a variety of teaching techniques and approaches, including problem-based learning, inquiry-based learning, and project-based learning. I also have experience in using technology, such as online learning platforms and educational software, to enhance the learning experience for my students.
In addition to my teaching experience, I have also completed advanced coursework in the subjects that I tutor, including mathematics, science, and language arts. This has allowed me to stay up-to-date on the latest educational trends and best practices, and to provide my students with the most current and effective teaching methods.
Overall, my hands-on experience and proficiency as a tutor have equipped me with the knowledge, skills, and expertise to help students achieve their academic goals and succeed in their studies.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Show how this diketone could be prepared by a condensation reaction:
-
The valuable polymer polyurethane is made by a condensation reaction of alcohols (ROH) with compounds that contain an isocyanate group (RNCO). Two reactions that can generate a urethane monomer are...
-
Show how 2-butanone could be prepared by a procedure in which all of the carbons originate in acetic acid (CH3CO2H).
-
Given an array, Arr[] of integer numbers have size N. The task is to find out single or consecutive numbers from Arr[] with 0 sum. When found, print "1" otherwise "0". Example-1: Input: 4 (6,-2,3,-1)...
-
4x 8 Describe the solution set as an inequality, in interval notation, and on a graph.
-
Clap Off Manufacturing uses 1,150 switch assemblies per week and then reorders another 1,150. If the relevant carrying cost per switch assembly is $5.50 and the fixed order cost is $625, is the...
-
What are the key elements of a project scope document?
-
No Doubt Company includes 1 coupon in each box of soap powder that it packs, and 10 coupons are redeemable for a premium (a kitchen utensil). In 2014, No Doubt Company purchased 8,800 premiums at 80...
-
Part of the Federal Agenda for Criminal Justice Reform proposed by members of Congress contains a proposal to incentivize states to reduce their prison populations. Enact the Reverse Mass...
-
8.a A Spartan missile and a Trojan missle are both flying at the s ame constant altitude. At time t, the Spartan missile is at the point: Clear[spartan, t] spartan[t_] = {16.1-7t+ t, 13 t - 2 t}...
-
For each of the following, identify the product (represented by A, B, C, and D) that would be formed through the indicated sequence of steps from the given starting material. O (1) mCPBA (2) EtMgBr...
-
Predict the major product from each of the following reactions. H O (1) (2) HO+ (3) HCrO4 MgBr (excess)
-
You work for LL Company, which manufactures high-end optical scopes for hunting rifles. LL Company has been the market leader for the past 20 years and has decided to diversify by applying its...
-
5. A 2.0 kg trolley moving east at 3.0 m/s collides head-on with a 1.0 kg trolley moving west at 2.0 m/s.. After the collision, the 2.0 kg trolley has a velocity of 1.0 m/s [E]. [4 marks] (a) What is...
-
20. For question 20, use the following quadratic function: f(x) = x-4x+4. A. Find the y-intercept, the equation of the axis of symmetry, and the x-coordinate of the vertex. y-intercept: axis of...
-
A simple harmonic oscillator consists of a mass 6.31 kg and an ideal spring with a linear restoring force. The oscillator's displacement is given by the equation x = 0.25 sin(5 t) where x is in...
-
Find the magnetic field (in milli-teslas, mT) that will produce a deflection of 0.30 radians for an electron beam in Thomson's experiment. The distance between the plates is 2.0 cm, the potential...
-
1. Two charges are set on the x -axis 9.90 cm away from each other. The charges are -8.40 nC and 18.3 nC. Calculate the electric potential at the point on the x-axis where the electric field due to...
-
What is the difference between pipelining and materialization?
-
The landing gear of an aircraft with: mass of 2000 kg the spring-mass-damper system Consider that the runway surface is y(t) = 0.2 cos 157.08t stiffness of the spring is 5 x 105 N/m. What is the...
-
What reagents and reaction conditions could be used to carry out the followingtransformations? Br - CH,CH,CH CH=CH, a) CH;CH,CH,CH;CH2 CH3 CH3 b) CH,CCI CH,COCH, CH3 CH3 C, Ph CH3 c) PHCH CI C=C CH3...
-
Suggest the best way to prepare these alkenes from alkylhalides. b) CH,CH CH,CH=CHCH_CH3 a) CH,CH,CH,CH,CH=CH, CH3 CH3 d)
-
When heated in ethanol, this alkyl halide gives two substitution and two elimination products. Show the structures of these products and the mechanism for theirformation. Br ELOH
-
New Flyer Industries has decided to expand its production of hybrid transit buses. The firm expects incremental cash flows of? $40 million per year for the next 10 years. The upfront cost of the...
-
Calculate the mean, mode, median, standard deviation, and range for the Graduation Rate, Salary After Completing Program, and Annual Cost. our response to each of these should include only the mean,...
-
1. Propel Corporation plans to make an investment, initially funded completely with debt. The investment requires an initial upfront cost of $49.8 million. The free cash flows of the investment and...

Study smarter with the SolutionInn App


