The Dow process for synthesizing phenol, which involves the reaction of chlorobenzene with aqueous sodium hydroxide at
Question:
The Dow process for synthesizing phenol, which involves the reaction of chlorobenzene with aqueous sodium hydroxide at high temperature and pressure, produces both diphenyl ether (1) and 4-hydroxybiphenyl (2) as by-products. Write mechanisms that account for the formation of 1 and 2.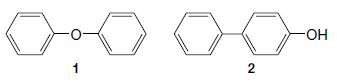
Transcribed Image Text:
1 2 -OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Solution The mechanisms can be written as follows Mechanism 1 Mechanism 1 the reaction of C6H5Cl wit...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
The hydrolysis of p-bromotoluene with aqueous sodium hydroxide at 300C yields m-methylphenol and p-methylphenol in a 5:4 ratio. What is the meta-para ratio for the same reaction carried out on...
-
Reaction of phenol with 1,2-epoxypropane in aqueous sodium hydroxide at 150C gives a single product, C9H12O2, in 90% yield. Suggest a reasonable structure for this compound.
-
Reaction of chlorobenzene with 4-chlorobenzyl chloride and aluminum chloride gave a mixture of two products in good yield (76%). What were these two products?
-
Suppose 2 x - T f(x) -1-cos z for all real numbers z. Find lim f(x) PIK 2) Use the Squeeze Theorem to evaluate the limit lim 2 sin()
-
The percentages of women and men who have high blood pressure are shown in Table 79 for various age groups. Let W and M be the percentages of women and men, respectively, at age a years who have high...
-
A three-phase motor is used to drive a pump. It is observed (by the use of a stroboscope) that the motor speed decreases from 898r/min when the pump is unloaded to 830 r/min as the pump is loaded. a....
-
A copy of the state of a database at a specific point in time during the middle of a normal business day is called a(n) a. archive c. checkpoint b. cookie d. restoration
-
Prepare, in good form, the 20X9 GAAP-based Statement of Revenues, Expenditures, and Changes in Fund Balance for the General Fund of Hicks Township, based on the following information: Property tax...
-
Cougar Inc. has the following standard and actual costs based on a a budgeted operating level of 10,000 units and an actual operating level of 10,500 units. Cougar Inc. Costs Cost Standard Actual...
-
The Bender Construction Co. is involved in constructing municipal buildings and other structures that are used primarily by city and state municipalities. This requires developing legal documents,...
-
For each of the following, identify the product (represented by A, B, C, and D) that would be formed through the indicated sequence of steps from the given starting material. (a) (b) (c) (d) CI (1)...
-
Write the mechanism for the following Heck reaction that can explain the final alkene stereochemistry and why the Pd species can promote the reaction to go to completion in only catalytic quantities....
-
During January 2019, Canyon Canoe Company completed the following nonmerchandising transactions: Jan. 2 Collected $4,500 on account. 15 Paid the utilities and telephone bills from December. 15 Paid...
-
Vera Spencer, a divorced mother of one, lived in a house she owned in suburban Mississauga, Ontario. Her daughter Karen was sixteen years old at the time of the events in question. Vera had an...
-
How would you write 139.29% in decimal form?
-
Addison sells bananas for $2.00 a bunch, and each bunch costs $1.50. By how much will Addison have to change the markup to match a competitor's selling price of $1.80 a bunch?
-
Simplify. 2 w+2w 15 2 6w -60w+126
-
Calculate the energy lost by an asteroid as it passed through the atmosphere If in space its mean velocity was 15 km/s, its diameter was 21 m and if the calculated kinetic energy needed to make the...
-
Refer to Apple's financial statements in Appendix A to answer the following. 1. For both fiscal years ended September 27, 2014, and September 28, 2013, identify the total amount of cash and cash...
-
Proposals have been made to ?sail? spacecraft to the outer solar system using the pressure of sunlight, or even to propel interstellar spacecraft with high-powered, Earth-based lasers. Sailing...
-
Show syntheses of these compounds from(Z)-2-butene: ) - a) H H C-C - CH3 . .
-
Show the products of thesereactions: . C-C CH3 1) O, CH,OH 1) O3. CH,OH a) CH;CH,CH-CH2 b) 2) (CH,),S 2) (CH,)2S CH3 . 1) 0,, CH;OH 1) 0,, CH,OH d) 2) (CH3),S 2) (CH3)2S 1) O3, CH,OH 2) (CH3),S
-
Show the alkenes that produce these compounds onozonolysis: a) CH,CCH,CH, + CH,CH b) c) CH;CCH,CH;CH + HCH + CH,CH
-
Problem 6-7 As the chief financial officer of Adirondack Designs, you have the following information: Next year's expected net income after tax but before new financing Sinking-fund payments due next...
-
LAW OF COMMERCIAL TRANSACTIONS Professor: Lois Yoder Beier Project 3: Wills, Trusts, and Estates and Investment Law. As their attorney, your clients. Frugal Fred and Barney Bizarre, now seek your...
-
An analyst has predicted the following returns for Stock A and Stock B in three possible states of the economy. State Boom Probability A B 0.29 0.22 0.24 Normal 0.52 0.15 0.16 Recession ? 0.09 0.08...

Study smarter with the SolutionInn App


