Write the mechanism for the following Heck reaction that can explain the final alkene stereochemistry and why
Question:
Write the mechanism for the following Heck reaction that can explain the final alkene stereochemistry and why the Pd species can promote the reaction to go to completion in only catalytic quantities.
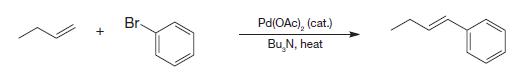
Transcribed Image Text:
+ Br- Pd(OAc), (cat.) Bu N, heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Solution The mechanism for the Heck reaction is as follows Proposed final alkene stereochemistr...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Write the mechanism for the reaction of a cysteine residue with iodoacetic acid.
-
Write the mechanism for the formation of carbon tetrachloride, CCI4, from the reaction of methane with CI2 + hv.
-
Write the mechanism for the base-catalyzed formation of a cyclic ester from a 1,7-diester.
-
Jilmar Company acquired a delivery truck and made payment of P2,870,000 analyzed as follows: Price of truck - P2,500,000 Charge for extra equipment - P150,000 Value added tax (recoverable) -...
-
The mean amounts of savings various age groups believe are enough to have at retirement are shown in Table 78 Let s be the mean amounts of savings (in millions of dollars) a person at age a years...
-
A six-pole, 60-Hz synchronous machine has a rotor winding with a total of 138 series turns and a winding factor kr = 0.935. The rotor length is 1.97 m, the rotor radius is 58 cm, and the air-gap...
-
Which backup method is the fastest? a. full daily backup c. differential daily backup b. incremental daily backup d. complete daily backup
-
Even with all of Marriotts brands they still do not have any hotel properties in the truly budget category of lodging. Thats a category occupied by properties such as Days Inn and Budgetel. Why do...
-
In 3 years Harry and Sally would like to have $18,000.00 for a down payment on a house. How much should they deposit each month into an account paying 6% compounded monthly? Payment = $
-
The heat flux that is applied to one face of a plane wall is q" = 20W/m. The opposite face is exposed to air at temperature 30C, %3D with a convection heat transfer coefficient of 20W/m2 . K. The...
-
The Dow process for synthesizing phenol, which involves the reaction of chlorobenzene with aqueous sodium hydroxide at high temperature and pressure, produces both diphenyl ether (1) and...
-
What are compounds A and B? Compound B has a strong IR absorption band in the 16501730 cm 1 region and a broad strong band in the 32003550 cm 1 region. 1-Methylcyclohexene (1) OSO (2) NaHSO A...
-
When acetaldehyde is treated with aqueous acid, an aldol reaction can occur. In other words, aldol reactions can also occur in acidic conditions, although the intermediate is different than the...
-
Roddy is CEO of Miss Pageants, Inc., a corporation organized for the purpose of planning and conducting beauty pageants. Roddy decided he wanted supermodel Winnie Harlow for mistress of ceremonies...
-
On the subject of US Economy following the pandemic: Describe the difference between financial assets and real assets and how that difference is particularly important when considering policy...
-
Task A: 1. Configure the network for the ABC College. a. Configure the PCs with the following information: Introduction: Assume yourself as a system administrator in ABC College. The college has...
-
b. Tony's Restaurant accepted a Visa card in payment of a $300 lunch bill. The bank charges a 2% fee. What entry should Tony's make? Sunland Company sold its accounts receivable of $81,000. What...
-
Can we treat the center of the Sun as a classical ideal gas? Let's find out. (a) Consider a gas at temperature T composed of particles of mass m. Use dimen- sional analysis to estimate the typical...
-
Santana Rey receives the March bank statement for Business Solutions on April 11, 2017. The March 31 bank statement shows an ending cash balance of $67,566. A comparison of the bank statement with...
-
A spacecraft has left the earth and is moving toward Mars. An observer on the earth finds that, relative to measurements made when the spacecraft was at rest, its a. length is shorter b. KE is less...
-
Show the products of thesereactions: H2 b) a) Pd Pt 2 . IH2 c) CH;C=CCH,CH; d) CH;C=CCH,CH3 Lindlar catalyst Pd 2 H2 Pt
-
Show all of the steps in the mechanism for this reaction and explain the regiochemistry of theaddition: Br Br + HBr
-
Show the products of thesereactions: HCI . H,SO, b) a)
-
BigScreen Corporation manufactures and sells televisions. Actual data relating to January, February, and March of 2 0 2 0 are: It is now April 2 0 2 0 and BigScreen takes a look back on the actual...
-
Consider the following three stocks: Stock A is expected to provide a dividend of $11.10 a share forever. Stock B is expected to pay a dividend of $6.10 next year. Thereafter, dividend growth is...
-
Determine the probability none of the four diamonds failing inspection. Place your calculation in cell C6. Use the BINOM.DIST function. Reference the value for the number of failures (B6) and the...

Study smarter with the SolutionInn App


