Using the (E )(Z ) designation [and in parts (e) and (f) the (R )(S ) designation
Question:
Using the (E )–(Z ) designation [and in parts (e) and (f) the (R )–(S ) designation as well give IUPAC names for each of the following:
(a)
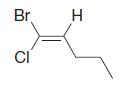
(b)
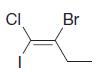
(c)
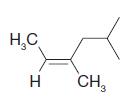
(d)
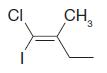
(e)
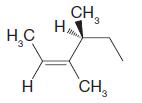
(f)
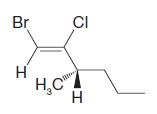
Transcribed Image Text:
Br H CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
If the two groups with the higher priorities are on the same ...View the full answer

Answered By

Hardik Dudhat
I am semi-qualified Chemical Engineering ,I have scored centum in accounting in my senior secondary and in my graduation. I have always helped my fellow students with their concerns on the subject, i have tutored on various tutoring sites in the past and also have taken home tuitions for degree and MBA students. As a tutor, I don't want my students to just get a solution, I want them to understand the concept and never have a doubt in that area thereon and i believe in excelling and not in educating.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Give the IUPAC names for each of the following compounds: (a) (CH3CH2)2C==CHCH3 (b) (CH3CH2)2C==C(CH2CH3)2 (c) (CH3)3CCH==CCl2 (d) (e) (f) (g) , ,
-
Give the IUPAC names for each of the following compounds: (a) CH2CH(CH2)5CHCH2 (b) (c) (CH2CH)3CH (d) (e) (f) CH2CCHCHCHCH3 (g) (h) CH3 CHz CH3 HH CI CI H H H3C CH,CH2 CH2CH3
-
Give the IUPAC names for each of the following compounds: (a) (CH3CH2)2C = CHCH3 (b) (CH3CH2)2C = C (CH2CH3)2 (c) (CH3)3CCH = CCl2
-
In Exercises 7-9, find the indicated measure. The area of a circle is 380 square inches. Find the radius.
-
Distinguish between a laissez-faire economist and an activist economist.
-
Why is the business entity assumption important?
-
On January 1st Mammoth Company established Juno Inc., a wholly-owned, self-sustaining British subsidiary with assets valued at 50 million pounds. The pound is the functional currency for Juno. The...
-
On the island of Pago-Pago, there are two lakes and 20 fishers. Each fisher gets to fish on either lake and gets to keep the average catch on that lake. On Lake X, the total number of fish caught is...
-
theoretical framework ( the determinant of the decision invest in fund) please provide reference
-
Use the following information to complete Phillip and Claire Dunphys 2019 federal income tax return. If any information is missing, use reasonable assumptions to fill in the gaps. Ignore the...
-
For the common-base network of Fig. 5.187 : a. Determine Z i and Z o . b. Calculate A v and A i . c. Determine a, b, r e , and r o . Fig. 5.187 hp =-0.992 h = 9.45 Q hob = 1 A/V 10 F 10 F 1.2 k2 2.7...
-
Write the structure(s) of the major product(s) obtained when 2-chloro-2,3-dimethylbutane reacts with (a) Sodium ethoxide (EtONa) in ethanol (EtOH) at 80 C or (in a separate reaction) with (b)...
-
The following information is available for the Albert and Allison Gaytor family in addition to that provided in Chapters 17. Albert sold the following securities during the year and received a Form...
-
Misstatements that are not corrected in a given year because they are not deemed material likely will affect the financial statements in the following year. This impact might be in the form of a...
-
True or False. \([A] \vec{X}=\lambda[B] \vec{X}\) is a standard eigenvalue problem.
-
True or False. The free-vibration equations corresponding to the transverse motion of a string, the longitudinal motion of a bar, and the torsional motion of a shaft have the same form.
-
What is Rayleigh's principle?
-
State the possible boundary conditions at the ends of a string.
-
Your client, Jacob, turned 66 years old this year. Jacob has no heirs and has decided that he would like to sell a life insurance policy to fund a trip to Africa that he has wanted to take. Jacob...
-
The percentage of completion and completed contract methods are described in the FASB ASC. Search the codification to find the paragraphs covering these topics, cite them, and copy the results.
-
Discuss the actual structure and the amount of resonance stabilization for the examples shown in problem 3.14.
-
Show the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. ) d) . CH3 H- b) CH3-N + H -H...
-
Draw the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. Discuss the relative...
-
38:38 k Problem 11-01 (algo, with video solution) Arjay purchases a bond, newly issued by Amalgamated Corporation, for $5,000. The bond pays $200 to its holder at the end of the first few years and...
-
=According to guidance in the IASB Codification, IASB 2 - Inventories, how should one account inventory costs using their cost formulas? How is this different from the FASB Codification (please...
-
Discuss how individuals' attitudes toward work have changed as a result of government programs and society's expectations.

Study smarter with the SolutionInn App


