What is the product of each of the following reactions? (a) (b) (c) (d) SnBu3 Pd catalyst
Question:
What is the product of each of the following reactions?
(a)
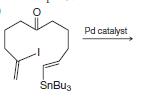
(b)
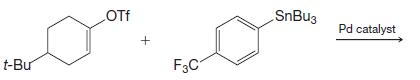
(c)
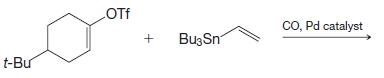
(d)
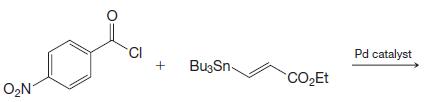
Transcribed Image Text:
SnBu3 Pd catalyst
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a The alkenyl iodide reacts with the organotin group at the other end of the ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
What is the product of each of the following reactions? a. ZnCl2 + CH3OH b. FeBr3 + Br- c. AlCl3 + Cl- d. + CH BF3 + HH
-
What is the product of the incidence matrix and its transpose for an undirected graph?
-
Give the major product of each of the following reactions a. b. c. d. e. f. g. h. i. CCH3 + HNO3 S NO2 CHs CH CHCH NCH CH 3 CH3 HO CH3NCH3 + PC15- NC 1. H202 2. CH3 1. HO 3. Ht CH3 + CH:CH2MgBr 2.4...
-
Simplify the following expressions according to the commutative law: a. b. A B + A C + B A c. (L M N)(A B)(C D E)(M N L) d. F (K + R) + S V + W + V S + W + (R + K) F A.B + B.A + C.D.E + C.DE + E.T.D
-
President Obamas statewide approval ratings and Democratic candidates shares of votes in Senate races in 2010 and 2012 are compared in Table 65 for 12 randomly selected Senate races in recent years....
-
Reporting of Notes Receivable, Interest, and Sale of Receivables On July 1, 2010, Mores an Company sold special-order merchandise on credit and received in return an interest-bearing note receivable...
-
Errors in mass transfer coefficients obviously affect the value of \(\mathrm{H}_{\mathrm{G}}\) and hence the height of the packed section. These errors also affect calculation of...
-
On November 1, 201X, Barbie Riley opened Barbies Art Studio. The following transactions occurred in November. 201X Nov. 1 Barbie Riley invested $8,000 in the art studio. 1 Paid ten months rent in...
-
1 . 5 . Select the option that is TRUE regarding the dividend growth model.a ) The dividend growth model is based on the principle that investors in ordinary sharesshould be rewarded for the risk...
-
The info following the questions are from the original example. Change the total fixed manufacturing overhead cost for the Milling Department in the area back to $390,000, keeping all of the other...
-
Provide the products of each of the following reactions. (a) (b) Ho 6 H+ Br- OH Cul, Pd catalyst Base (an amine)
-
What starting materials could be used to synthesize the following compound by a SuzukiMiyaura coupling? H.
-
A researcher claims that the number of homicide crimes in California by month is uniformly distributed. To test this claim, you randomly select 1800 homicides from a recent year and record the month...
-
Consider the two small, equal-mass, charged spheres shown in the figure. The top sphere is suspended from the ceiling by a filament, and has a charge of q = 28.0 nC. The bottom sphere has a charge of...
-
11. Write a program named Weather Report that uses arrays: a. Compute the average temperature for that week. b. The number of days for which the temperature was taken. c. How many days temperature...
-
What happens to the pH of an acid as water is added? Give a specific example where this readout will knowingly be wrong. Explain.
-
If f(x) = x+5, g(x) = x 4, h(x) = , then fogo h(x)=
-
Using the replacement cost method, calculate the actual cash value of a roof that was destroyed 2 0 years after the purchase of it . The roof has a 2 5 - year life span and the insurance company has...
-
1. At year-end, Harris Company had shipped $12,500 of merchandise FOB destination to Harlow Co. Which company should include the $12,500 of merchandise in transit as part of its year-end inventory?...
-
For all of the following words, if you move the first letter to the end of the word, and then spell the result backwards, you will get the original word: banana dresser grammar potato revive uneven...
-
Calculate these quantities: (a) The wavelength of light (in centimeters) with a frequency of 9.00 x 1012Hz. (b) The frequency of light with a wavelength of 310nm (c) The energy of light (in kcal/mol...
-
What kind of light has a frequency of 9.00 x 1013Hz?
-
Some NMR spectrometers operate at 4 x 108Hz (400MHz), what is the energy of this radiation?
-
QUESTION 28 Given the following information, calculate the operating expense ratio for this property: potential gross income: $245,000; vacancy rate: 9%; ne operating income: $57,900; operating...
-
24. Which one of the following will increase the return on equity as computed using the Du Pont identity given that all else is held constant? A. A decrease in sales B. A decrease in net income C. An...
-
Paddleboard Inc. began operations on January 1 , 2 0 2 2 . Its post - closing trial balance at December 3 1 , 2 0 2 2 and 2 0 2 3 , is shown below along with some other information. [ [ [ [...

Study smarter with the SolutionInn App


