What starting materials could be used to synthesize the following compound by a SuzukiMiyaura coupling? H.
Question:
What starting materials could be used to synthesize the following compound by a Suzuki–Miyaura coupling?
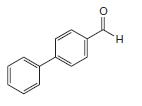
Transcribed Image Text:
H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Solution The starting materials for this method are an olefin a boronic acid and copper II c...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
What starting materials could be used to synthesize each of the following compounds by a Sonogashira coupling reaction? (a) (b) CI
-
Synthesize the following compound starting with ethyne and 1-bromopentane as your only organic reagents (except for solvents) and using any needed inorganic compounds. Br Br
-
Synthesize the following compound by a method that derives all carbon atoms from alcohols of five carbons or fewer. You may use any other reagents needed. OH
-
A copper-zinc alloy has the following properties: Grain diameter (mm) Strength (MPa) 0.015 .................................. 170 MPa 0.025 .................................. 158 MPa 0.035...
-
The windchill (or windchill factor) is a measure of how cold you feel as a result of being exposed to wind. Table 64 pro- vides some data on windchills for various temperatures when the wind speed is...
-
Financial Statement Presentation of Receivables Patriot Company shows a balance of $241,140 in the Accounts Receivable account on December 31, 2010. The balance consists of the following. Illustrate...
-
Although the largest errors in calculating the height of a packed column are errors in (1) mass transfer coefficients and (2) VLE data, calculation errors can also be significant because calculation...
-
An alphabetical list presenting the accounts and balances for Kens Cleaners on June 30, 2014, follows. All the accounts have normal balances. Accounts Payable .............. $ 30,840 Accounts...
-
A Treasury bond that matures in 1 0 years has a yield of 5 . 5 0 % . A 1 0 - year corporate bond has a yield of 8 . 2 5 % . Assume that the liquidity premium on the corporate bond is 0 . 3 5 % . What...
-
During the month of June, Bon Voyage Travel recorded the following transactions: 1. Owners invested $25,000 in cash to start the business. They received common stock. 2. The month's rent of $500 was...
-
What is the product of each of the following reactions? (a) (b) (c) (d) SnBu3 Pd catalyst
-
Starting with 1-butanol and using any other required reagents, outline a synthesis of each of the following compounds. You need not repeat steps carried out in earlier parts of this problem. (a)...
-
What are three characteristics that make a given stimulus more likely to be selected for attention?
-
Discussion | Catalina The newest version of the Mac operating system, macOS 10.15 (Catalina), was released in Fall 2019. Read the article below and in your own sentences summarize the major updates...
-
Chen Lee is a 44-year-old Chinese citizen who lives and works in Shanghai, China. He is divorced and has the custody of his two boys: Wang Lee, who is 15 years old, and Lian Lee, who is 19 years old....
-
ANALYZE THE RELEVANT ENVIRONMENT that you could face in your chosen career/industry and 4. Carefully describe your PERSONAL ETHICAL STATEMENT (see AMA Code) that will guide your decision making as a...
-
If the total obligation is $950 and borrower income is $4,500, the total obligation ratio is?
-
Mr. Morani doesn't fall under the provisions of A112(2), or A112(3), and is eligible to apply for PRRA. Mr. Morani has no previous refugee or PRRA claims, so he could present all the personal and...
-
Refer to Apple's financial statements in Appendix A to answer the following. Required 1. Assume that the amounts reported for inventories and cost of sales reflect items purchased in a form ready for...
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
Explain which compound has the higher melting point.
-
Explain which compound has the higher boiling point. Discuss in detail.
-
Explain whether each compound is soluble in aqueous NaOH, aqueous NaHCO3 both, or neither.
-
Hi this is a question about business finance, thank you!
-
You interviewed a new client and these were the facts provided. Provide them with a brief explanation of tax reason for each Their home mortgage interest is $ 1 2 0 0 , real estate taxes $ 8 , 2 0 0...
-
Part A Capital Budgeting Background: Electro Motors (Electro) is considering a new project to produce electric vehicles for the Australian domestic market and international markets. It has identified...

Study smarter with the SolutionInn App


