Alkenes undergo reaction with peroxycarboxylic acids to give epoxides. For example, cis-but-2-ene gives 2,3-epoxybutane: H3C
Question:
Alkenes undergo reaction with peroxycarboxylic acids to give epoxides. For example, cis-but-2-ene gives 2,3-epoxybutane:
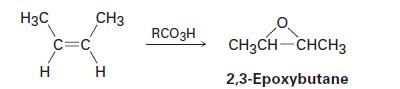
Transcribed Image Text:
H3C Н Н C=C CH3 Н RCO3H CH3CH-CHCH3 2,3-Epoxybutane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Thats correct Alkenes can undergo a reaction with peroxycarboxylic acids to form epoxides a t...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structures of all epoxides that could in principle be formed when each of the following alkenes reacts with meta-chloroperoxybenzorc acid (mCPBA). Which epoxide should predominate in each...
-
Alkenes slowly undergo a reaction in air called autoxidation in which allylic hydroperoxides are formed. OOH Cyclohexene Oxygen 3-Hydroperoxycyclohexene 0:0)
-
Give the structures of the two stereoisomeric alkenes with the molecular formula C6H12 that react with HI to give the same single product and undergo catalytic hydrogenation to give hexane.
-
Perform the indicated operations and then simplify. Assume that all variables represent nonzero real numbers. 20yx3 + 15y4x + 25yx4 10yx
-
You are a bidder in an independent private values auction, and you value the object at $ 4,000. Each bidder perceives that valuations are uniformly distributed between $ 1,500 and $ 9,000. Determine...
-
In Example 11.4 (Figure 11.13), assume the round trip time for a frame is 40 milliseconds. Explain what will happen if we set the time-out in each of the following cases. Figure 11.13 a. 35...
-
In the months leading up to the 2016 election, Christopher Steele, a former British intelligence agent, was hired by a Washington, D.C., research firm to investigate whether then-candidate Donald...
-
The partners in New Yorker Company decide to liquidate the firm when the balance sheet shows the following The partners share income and loss 5 : 3 : 2. During the process of liquidation, the...
-
2. In the system below, the coefficient of friction between the block and the table is 0.2. T2 2 kg 5 kg Find the acceleration of the system and the tensions T1 and T2. 10 kg [8]
-
We said in Section 4.9 that an allylic carbocation is stabilized by resonance. Draw resonance structures to account for the similar stabilization of a benzylic carbocation. + CH A benzylic carbocation
-
Draw the two cistrans stereoisomers of 1,2 dimethyl cyclopentane, assign R,S configurations to the chirality centers, and indicate whether the stereoisomers are chiral or meso.
-
We have been requested by a large retailer to submit a bid for a new point-of-sale credit checking system. The system would be installed, by us, in 89 stores per year for three years. We would need...
-
Taras Textiles currently has credit sales of $30 million per month, an average collection period of 60 days, and bad debts equal to 3% of sales. Assume that the price of Taras products is $60 per...
-
Describe the two ways the BLS measures total employment.
-
On Friday, August 28, 2020, the stock of Tesla Inc. closed at $2,213.40 per share. On the following Monday, the company split its stock 5-for-1, and the stock price traded for $444.61 per share. Were...
-
U.S. companies had been cutting back on their working capital investments, most recently by dialing back inventories. Suppose that firms are managing their working capital assets to maximize...
-
What are the various ways in which the Federal Reserve can influence the money supply?
-
Jenny Smith owns Classic Photography Inc., a company that restores photos for its clients and creates electronic images from the restored photos. The company also frames restored photos and creates...
-
A heat engine has a heat input of 3 Ã 104 Btu/h and a thermal efficiency of 40 percent. Calculate the power it will produce, in hp. Source 3 x 10 Btu/h 40% HE Sink
-
Consider the structure of N,N-dimethylformamide (DMF): We might expect the two methyl groups to be equivalent; however, both the proton and carbon NMR spectra of DMF show two separate signals for the...
-
Consider the structure of phenol: The chemical shift of the hydroxyl proton is found to be sensitive to the concentration of phenol. In a concentrated solution, the hydroxyl proton produces a signal...
-
Consider the two methyl groups shown in the following compound. Explain why the methyl group on the right side appears at lower chemical shift. CH3 0.8 ppm 1.0 ppm H;C.
-
a) Suppose you have the following quotations: /$ /$ 0.8032 -0.8048 125.000 -125.110 What are the bid-ask prices of the yen in euro or / ? [15%] b) Ingo Schmidt, a cross-rate trader at Deutsche Bank,...
-
Taussig Technologies Corporation (TTC) has been growing at a rate of 20% per year in recent years. This same growth rate is expected to last for another 2 years, then decline to g n = 8%. If D 0 =...
-
Grey Sky Construction needs a piece of equipment that costs $60,000. Grey Sky either can lease the equipment for two years at $31,000 per year, paid at the beginning of the year, or borrow $60,000...

Study smarter with the SolutionInn App


