E or Z stereochemistry to the following compounds: (a) H3C C=C CH3CH2 CHOH CI (b) CI CH30
Question:
E or Z stereochemistry to the following compounds: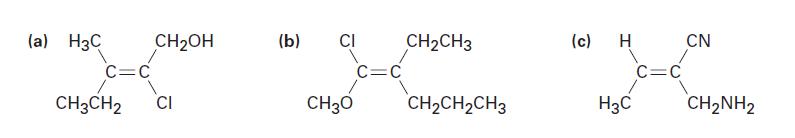
Transcribed Image Text:
(a) H3C C=C CH3CH2 CH₂OH CI (b) CI CH30 C=C CH₂CH3 CH₂CH₂CH3 (c) H H3C C=C CN CH₂NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To determine the E trans or Z cis stereochemistry of the given compounds we need to look at the subs...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give structures (including stereochemistry where appropriate) for compounds A-G: (a) (b) (c) (d) 2 NaNH2 Benzene+ o c heat (1) Li, EtNH2 E (C9H10 (2) NHCI (Section 7.15B)) Br2 D F+ enantiomer (major...
-
Write structures for compounds A-J showing stereochemistry where appropriate. (a) What is the stereochemical relationship between A and C? (b) (c) What is the stereochemical relationship between H...
-
Compounds A through H are isomeric bromoalkanes with the molecular formula C5H11br. With your team, draw all eight constitutional isomers. Indicate any stereo-center) (s), but do not label it (them)...
-
Write code in MATLAB (Radionuclide) (half-life) U-238 4.468 x 10 years U-235 703.8 x 10 years Mo-99 67 hours Tc-99m 6.04 hours Given the formula, where is the decay constant used in , calculate the...
-
The Inventory account for Dennis Handy Man Company had a balance of $1 32,100 at the end of its fiscal year. A physical count taken at year end revealed that the value of inventory on hand amounted...
-
For the classical normal regression model y = X + with no constant term and K regressors, what is plim F[K, n K] = plim R 2 /K / (1R 2 )/(nK), assuming that the true value of is zero?
-
Identify the most appropriate approach and then design. Provide a rationale as to why this approach and design would be most appropriate. Develop a hypothetical research scenario that would...
-
A comparative balance sheet for Orozco Corporation is presented below. Additional information:1. Net income for 2012 was $105,000.2. Cash dividends of $40,000 were declared and paid.3. Bonds payable...
-
(20%) The output of an argon ion laser can consist of a number of modes of frequency that match the cavity resonance condition and are within the gain bandwidth of the lasing transition. Assume the...
-
Change the following old names to new, post-1993 names, and draw the structure of each compound: (a) 2,5,5-Trimethyl-2-hexene (b) 2,2-Dimethyl-3-hexyne (c) 2-Methyl-2,5-heptadiene (d)...
-
What are the likely formulas of the following molecules? (a) AlCl? (b) CF2Cl? (c) NI? (d) CH?O
-
Disarmed retroviral vectors can be used to introduce genes into higher animals including humans. What advantages do retroviral vectors have over other kinds of gene-transfer vectors? What...
-
Karen Klaus has a business building (Section 1250 property) with an adjusted basis of $40,000 and a fair market value of $160,000. The building originally cost $170,000 when purchased in 2015. In...
-
Steven Stronghold had a Section 1231 net gain of $25,000 in 2019. His previous net Section 1231 items were $12,000 in 2012, ($8,000) in 2013, ($6,000) in 2014, ($3,000) in 2015, ($1,000) in 2016,...
-
$20 million of nondealer real estate obligations arose in and are outstanding at the end of calendar-year 2019. Deferred gross profit on such installment obligations equals $5 million. Assume that...
-
Assume the facts of the preceding problem except that the building was an apartment building. a. How much income does Ivan recognize and how will it be treated? b. What would your answer be to (a) if...
-
Marielle Machinery Works is considering a project which has an initial investment of 10,000 and has expected cash flows of 0 in year 1, 7,500 in year 2, and 8,500 in year 3. The company uses the IRR...
-
With corporate offices in Quebec, RONA Inc. operates approximately 800 stores of various sizes and format across Canada and is the largest Canadian distributor and retailer of hardware, renovation,...
-
Find the reduced echelon form of each of the matrices given in Problems 120. c 1 26 + 4
-
A chiral sample gives a rotation that is close to 180. How can one tell whether this rotation is +180 or -180?
-
Convert the following perspective formulas to Fischer projections. (a) (b) (c) (d) H OH .C. CH; CH,OH
-
Convert the following Fischer projections to perspective formulas. (a) (b) (c) (d) H,N- H- CH;
-
Write a make a reflective journal on What is Organizational Behavior and Management. By including these points . with reference and 500 words 1. Network with participants in the unit 2. Define...
-
The case focuses on QANTAS, and the role of Olivia Wirth (executive of Government and Corporate Affairs) who has to respond to the company's failed and publicly scrutinised social media contest...
-
Total of BS of Assets side in question given $82,000 while it comes $82,200 No repayment of loan and payment of interest. 1 Step 1 Prepare a Purchase Budget First Store Purchase Budget For the...

Study smarter with the SolutionInn App


