Name the following alkyl halides: (a) H3C Br Br | | || CH3CHCHCHCHCHCH3 CH3 (d) CHBr CH3CHCHCHCHCH3
Question:
Name the following alkyl halides:
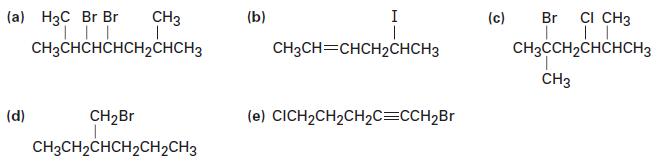
Transcribed Image Text:
(a) H3C Br Br | | || CH3CHCHCHCH₂CHCH3 CH3 (d) CH₂Br CH3CH₂CHCH₂CH₂CH3 (b) I CH3CH=CHCH2CHCH3 (e) CICH₂CH₂CH₂C=CCH₂Br (c) Br CI CH3 | || CH3CCH₂CHCHCH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
a 1bromo22dimethylpropane b 2bromo3methyl pentane c 3bromo1chlorobutane d 2bromo...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give a JUPAC name for each of the following alkyl halides (yellow green =Cl): (b) (a)
-
Which of the following alkyl halides form a substitution product in an SN1 reaction that is different from the substitution product formed in an SN2 reaction? a. b. c. d. e. f. CH Br CH CHCHCHCH CHa...
-
Tell which of the following alkyl halides can give only one alkene, and which can give a mixture of alkenes, in the E2 reaction. (a) (b) CH,CH,CHCH,Br CH3
-
Discuss why the length of an OH bond obtained from X-ray diffraction experiments averages 85 pm whereas that obtained in neutron diffraction experiments averages 96 pm. Would you expect to see...
-
Describe how a manager who derives satisfaction from both income and shirking allocates a 10- hour day between these activities when paid an annual, fixed salary of $ 100,000. When this same manager...
-
What are three important differences between the 3G and 4G cellular architectures?
-
This case deals with several issues regarding contract formation under the UCC. Logan and Kanawha Coal agreed to purchase coal from Detherage via a fax dated March 9, 2010. The fax stated that it had...
-
Develop an opportunity loss table for the investment problem that Mickey Lawson faces in Problem 3-20. What decision would minimize the expected opportunity loss? What is the minimum EOL?
-
What is Data Manipulation Language in SQL ? What are the Benefits of DML? What is DML command with example?
-
Draw structures corresponding to the following names: (a) 2-Chloro-3,3-dimethyl hexane (b) 3,3-Dichloro-2-methyl hexane (c) 3-Bromo-3-ethyl pentane (d) 2-Bromo-5-chloro-3-methylhexane
-
Ignoring double-bond stereochemistry, what products would you expect from elimination reactions of the following alkyl halides? (a) Br CH3 | | CH3CH2CHCHCH3 (b) CH3 CI CH3 | | CH3CHCH2-C-CHCH3 CH3...
-
A person walks in the following pattern: 3.1km north, then 2,4km west, and finally 5.2km south. (a) Sketch the vector diagram that represents this motion. (b) How far and (c) In what direction would...
-
What happens to the difference between ATC and AVC as a firms output expands? Explain.
-
If the rise in the price of service A leads to a fall in the price of service B, we may conclude that __________. a) services A and B are substitutes b) services A and B are complements c) services A...
-
Kirsten pays $2 for a bottle of water. Explain how its possible for her to be enjoying a consumer surplus.
-
Suppose the price of a service falls and people buy more of that service. What has happened? a) Quantity demanded changed. b) Demand increased. c) Demand decreased.
-
Which statement is false? a) The waterdiamond paradox can be resolved with the help of the law of diminishing marginal utility. b) We will consume a service when its marginal utility is equal to its...
-
There have been an increasing number of editorials in newspapers and magazines about their concern over transport capacity and infrastructure. What is the nature of this issue? Why is it such a...
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
For each pair of the following compounds identify which compound would react more rapidly in an E1 reaction. a. b. CI .CI CI CI
-
Identify the stronger base: a) NaOH vs. H 2 O b) Sodium ethoxide vs. ethanol c) Ammonia vs. trimethylamine
-
(2S, 3S)-2-Bromo-3-phenylbutane undergoes an E2 reaction when treated with a strong base to produce (E)-2- phenyl-2-butene. Use Newman projections to explain the stereo-chemical outcome of this...
-
Determine if the improper integral ( \int_{1}^{\infty} \frac{1}{x^2} dx ) converges or diverges. If it converges, calculate its value.
-
Find the volume of the solid formed when the region enclosed by the curves ( y = \sqrt{x} ), ( y = 0 ), and ( x = 4 ) is revolved around the ( y )-axis. Provide a detailed solution using the disk or...
-
Evaluate the definite integral ( \int_{0}^{2} (4x - x^2) dx ) and interpret the result as the area between the curve ( y = 4x - x^2 ) and the ( x )-axis over the interval ([0, 2]).

Study smarter with the SolutionInn App


