Give a JUPAC name for each of the following alkyl halides (yellow green =Cl): (b) (a)
Question:
Give a JUPAC name for each of the following alkyl halides (yellow green =Cl):
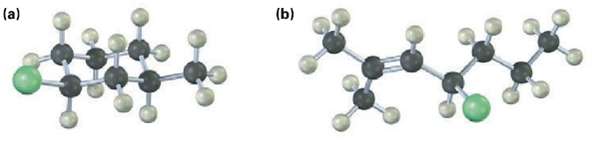
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a CH3 C CH3 CH3...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give the IUPAC name for each of the following alkyl groups, and classify each one as primary, secondary, or tertiary: (a) CH3(CH2)10CH2-- (b) (c) --C(CH2CH3)3 (d) (e) (f) -CH2CH2CHCH2CH2CH3 CH2CH3...
-
Give the IUPAC name for each of the following alkyl groups, and classify each one as primary, secondary, or tertiary: (a) CH3 (CH2)10CH2 (b) CH2CH2CHCH2CH2CH3 W CH2CH3
-
Give a substitutive name for each of the following compounds. (a) CH3CHT-O-CH'CH'-OH (b)
-
The MRP gross requirements for Item A are shown here for the next 10 weeks. Lead time for A is three weeks and setup cost is $ 10. There is a carrying cost of $ 0.01 per unit per week. Beginning...
-
Consider the stylized example of Figure 7.3, in which there is a jump in the mean value. Using the same worksheet as a template, enter the exponential smoothing calculations for these periods. Assume...
-
What is power? How is leadership different from power?
-
In its 2014 annual report to shareholders, Johnson & Johnson, a consumerproducts company, reported the following financial results: 1. Revenues increased from \($71.3\) to \($74.3\) billion. 2. Net...
-
The Ferre Publishing Company has three service departments and two operating departments. Selected data from a recent period on the five departments follow: The company allocates service department...
-
3w 20, y = 2x + 1/3-1
-
Use cost structure concepts (ie fixed costs, variable costs, semi variable costs mixed costs, and step costs step fixed costs) to answer the following questions Suppose the community college wants to...
-
Tell whether each of the following reactions is an oxidation, a reduction, orneither. (a) NABH4 H20 CH;CH- CH3CH2CH2OH (b) OH 1. BH3 2. NaOH, H202
-
Show the product(s) of reaction of the following alkenes withNBS: (b) (a)
-
(a) For the three degree-of-freedom system shown in Figure 6.82, derive the equations of motion and solve for the frequencies and modes of vibration for the arbitrary stiffnesses \(k_{1}, k_{2},...
-
1.Identifying and interpreting the information for diagnosis and repair of the air brake systems: You must identify three Best Practices and Methods for obtaining and Analysing information from each...
-
Why was opening trade with China permanently different from other trade agreements? What were its positive and negative impacts on the US?
-
An underground fire service main in a methanol plant consists of glass reinforced plastic material. The piping is rated for 200 psi. The pump's churn pressure is rated at 280 psi. Numerous leaks have...
-
A mechanic works in a shop that uses a flat rate pay system. The flat-rate time for replacing water pump is 2 and 6/10 hours and the flat rate pay is $60. He takes 2 and 1/10 hours to change the...
-
Consider the $1.90 per person per day as the relevant international poverty line and define an appropriate weekly poverty line. Compute the poverty severity index for this population of households....
-
Determine the frequency response of the system of Figure P6.8 and Chapter Problem 6.43 due to a sinusoidal force \(F_{0} \sin \omega t\) applied to the mass center of the machine tool. Data From...
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
The titration of 10.00 mL of HCl solution of unknown concentration requires 12.54 mL of a 0.100 M NaOH solution to reach the equivalence point. What is the concentration of the unknown HCl solution...
-
Construct a graph, similar to Figure 3-11, of the torsional energy of 3-methylpentane along the C2-C3 bond. Place C2 in front, represented by three bonds coming together in a Y shape, and C3 in back,...
-
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly. (a) 2-ethylpentane (b) 3-isopropylhexane (c)...
-
Provide IUPAC names for the following compounds. (a) (CH3)2CHCH2CH3 (b) CH3-C(CH3)2-CH3 (c) (d) (e) (f) CH CH CHCH CH,CHCHCH le ' CH CH,CH, CH CH CH, CH CH C(CH CH,CH,CHCHCH, CH(CH2 CH CHCH,CH, CH)C...
-
Consider the following information on a portfolio of three stocks: State of Probability of State Stock A Rate of Economy of Economy Return Stock B Rate of Return Stock C Rate of Return Boom Normal...
-
3 Self-assessment activity Consider the following two independent scenarios: 3.1. Complete the following table: Mark-up on Cost price Profit Selling price cost (Excl. VAT) (Excl. VAT) (Excl. VAT) 10%...
-
If you deposit $800 into a bank account today, what annually compounded interest rate would you need to earn in order to have $2,200 in 12 years? Enter your answer as a percentage rounded 2 decimal...

Study smarter with the SolutionInn App


