Propose structures for nitriles that might be precursors of the following amines: (a) CH3 T CH3CHCHCHNH (b)
Question:
Propose structures for nitriles that might be precursors of the following amines: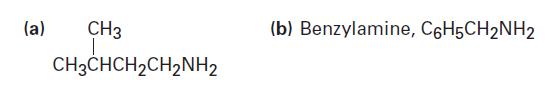
Transcribed Image Text:
(a) CH3 T CH3CHCH₂CH₂NH₂ (b) Benzylamine, C6H5CH2NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To propose structures for nitrile precursors of the given amines we need to iden...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose structures for amides that might be precursors of the following amines: (a) CH3CHCHNH2 (b) NH(CH2CH2CH3)2 (c) CHNH2
-
Show how you might prepare each of the following amines through reductive amination: (a) (b) (c) NH2 CH3 CH3
-
How might the following amines be prepared using reductive amination reactions? Show all precursors if more than one ispossible. CH (b) NHCH2CH3 (c) (a) CHCH2NHCHH -NHCH3
-
In Exercises 8194, begin by graphing the absolute value function, f(x) = |x|. Then use transformations of this graph to graph the given function. h(x) = x + 4 - 2
-
Finx, Inc., purchased a truck for $35,000. The truck is expected to be driven 15,000 miles per year over a five-year period and then sold for approximately $5,000. Determine depreciation for the...
-
Describe an example of a text T of length n and a pattern P of length m such that the brute-force pattern-matching algorithm achieves a running time that is (nm).
-
Alternatives 1, 2, and 3 have lives of 3, 4, and 6 years, respectively. Their net cash flow (NCF) and salvage value (SV) profiles are as follows: Additional explanation is necessary: The NCF profile...
-
Pro Tire manufactures tires for all- terrain vehicles. Pro Tire uses job costing and has a perpetual inventory system. On November 22, Pro Tire received an order for 190 TX tires from ATV Corporation...
-
1)Consider the crowding out model. Assume there are men and women in the labor force and three occupations: preschool, elementary and secondary teachers. Suppose only women can work as elementary and...
-
The , to , interconversion of unsaturated ketones is catalyzed by base as well as by acid. Propose a mechanism. Problem 11.29 Nonconjugated , -unsaturated ketones such as cyclohex-3-enone are in an...
-
Classify each of the following compounds as either a primary, secondary, or tertiary amine: (a) CH3 CH3CH2CHNH2 (b) N-H (c) -N CH3 CH3
-
Kevlar, a nylon polymer prepared by reaction of 1, 4-benzenedicarboxylic acid (Terephthalic acid) with 1, 4-benzenediamine (p-phenylendiamine), is so strong that its used to make bulletproof vests....
-
Thomas transfers land worth $100,000 with an adjusted basis of $35,000 and a mortgage of $65,000 and equipment worth $30,000 and a basis of $10,000 to Andy Co. in return for 50 shares of stock....
-
Prepare an exponential smoothing forecast.
-
Prepare a weighted-average forecast.
-
Prepare a linear trend forecast.
-
Construct control charts and use them to monitor forecast errors.
-
Are budgets typically recorded in Debt service funds?
-
Problem 3.5 (4 points). We will prove, in steps, that rank (L) = rank(LT) for any LE Rnxm (a) Prove that rank (L) = rank (LTL). (Hint: use Problem 3.4.) (b) Use part (a) to deduce that that rank(L) =...
-
Determine the electron configuration for each of the following atoms: a. Carbon b. Oxygen c. Boron d. Fluorine e. Sodium f. Aluminum
-
In each case, identify the more stable anion. Explain why it is more stable. (a) (b) (c) vs. N. vs. -zo
-
Atropine, extracted from the plant Atropa belladonna, has been used in the treatment of bradycardia (low heart rate) and cardiac arrest. Draw the enantiomer of atropine: CH 0= -
-
2) Steam enters an adiabatic turbine at 500 psia and 900 F at a flow rate of 480 ft/min. Expansion in the turbine leads to an exit pressure of 20 psia. Calculate the turbine power output (in hp) if...
-
15) Radiation to a Tube from a Large Enclosure. A small oxidized horizontal metal tube with an OD of 0.0254 m (1-inch), 0.61 m (2 ft) long, and with a surface temperature at 472 K (390 F) is in a...
-
1) A 20-mm-diameter steel (E = 200 GPa, v = 0.28) tension rod is 2 meter long and carries a load of 4500 N. a. Calculate tensile stress (MPa) b. Calculate tensile strain c. Change in length of the...

Study smarter with the SolutionInn App


