Tert-Butyl ethers react with trifl uoroacetic acid, CF 3 CO 2 H, to yield an alcohol and
Question:
Tert-Butyl ethers react with trifl uoroacetic acid, CF3CO2H, to yield an alcohol and 2-methylpropene. Tell what kind of reaction is occurring, and propose a mechanism.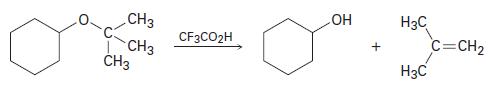
Transcribed Image Text:
CH3 CH3 CH3 CF3CO2H + H3C H3C C=CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The reaction of tertbutyl ethers with trifluoroacetic acid CF3CO2H to yield an alcohol and 2methylpr...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1, 3-Cyclopentadlene reacts with Cycloheptatrienone to give the product shown. Tell what kind of reaction is Involved, and explain the observed result. Is the reaction suprafacial orantarafacial?...
-
On standing, 1,3-cyclopentadiene is transformed into a new compound called dicyclopentadiene, having the molecular formula C10H12. Hydrogenation of dicyclopentadiene gives the compound shown. Suggest...
-
A study by the Pew Internet and American Life Project (pewinternet.org) found that Americans had a complex and ambivalent attitude toward technology. (Data extracted from M. Himowitz, "How to Tell...
-
What is project scoping? Why is it important to good problem solving?
-
The Hull Petroleum Company and Inverted V are retail gasoline franchises that compete in a local market to sell gasoline to consumers. Hull and Inverted V are located across the street from each...
-
Show how the Adler algorithm (Figure 10.19) attaches weights to the data items when calculating the checksum. Figure 10.19 Start Notes L: Left 16-bit checksum R: Right 16-bit checksum D;: Next 16-bit...
-
Almetals, Inc., a Michigan company, entered into a contract with the German firm Wickeder Westfalenstahl regarding the purchase of clad metal, a specialty metal used in a variety of industries but...
-
The segmented income statement for XYZ Company for the year ended December 31, 2010, follows: The company is concerned about the performance of product A, and you have been asked to analyze the...
-
(d) Suppose that instead of restricting imports or giving explicit advantages to Canadian-made products, the Bank of Canada enters the foreign exchange market and buys up large quantities of foreign...
-
How would you prepare the following compounds from 2-phenylethanol? (a) Benzoic acid (b) Ethylbenzene (c) 1-Bromo-2-phenylethane (d) Phenylacetic acid (C 6 H 5 CH 2 CO 2 H) (e) Phenylacetaldehyde (C...
-
But-2-ene-1-thiol is a component of skunk spray. How would you synthesize this substance from but-2-en-1-ol? From methyl but-2-enoate, CH 3 CH=CHCO 2 CH 3 ? More than one step is required in both...
-
A The owner of a small business selling and repairing cars which you patronise has just received a copy of his accounts for the current year. He is rather baffled by some of the items and as he...
-
In Heap-Sort once a root element has been put in its final position, how much time, does it take to re-heapify the array so that the next removal can take place? In other words, what is the Time...
-
Argentina, once the worlds seventh-largest economy, has long been considered one of Latin Americas worst basket cases. Starting with Juan Peron, who was first elected president in 1946, and for...
-
The stock of Darden Restaurants fell from $120.68 to $39 in just 22 days. Prior to the coronavirus outbreak, Darden was on pace to pay a dividend in 2020 of $3.52 per share. Given that dividend, and...
-
The worst-case runtime Complexity of a search of a value in a BST with n nodes is: O(n 2 ) O(n * log n) O(n) O(logn)
-
Ford exports SUVs from the United States to Europe in competition with the Japanese. Similarly, Apple exports computers to Europe. However, all of Apples biggest competitors are American...
-
Is bitcoin still a viable currency? Use an Internet search engine to nd out. If so, what is the exchange rate for one bitcoin in the currency of your country? Would you buy a bitcoin? If so, why? If...
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
Draw all possible conjugated dienes with molecular formula C 6 H 10 , taking special care not to draw the same compound twice.
-
Treatment of 1,2-dibromocycloheptane with excess potassium tert-butoxide yields a product that absorbs UV light. Identify the product.
-
In each of the following pairs of compounds identify the compound that liberates the most heat upon hydrogenation. (a) (b)
-
We all have our own biases that we may not even realize we have our implicit biases. Harvard's Project Implicit website provides a test that you can use to explore the implicit biases you may have....
-
Air expands through a turbine from 7bar, 960 K to 1.0 bar, 450 K. The inlet velocity is small compared to the exit velocity of 75 m/s. The turbine operates at steady state and develops a power output...
-
Nauto, a data platform for self-driving cars, helps make autonomous driving safer for companies managing fleets of self-driving vehicles. Naturally, its customers would need all kinds of information,...

Study smarter with the SolutionInn App


