The following structure represents a carbocation. Draw two resonance structures, indicating the positions of the double bonds.
Question:
The following structure represents a carbocation. Draw two resonance structures, indicating the positions of the double bonds.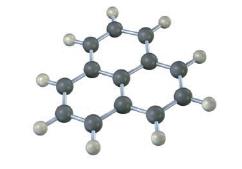
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
Our task here is to draw two additional resonance structures of the molecule given below First we wi...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw two resonance structures for diazomethane, CH2N2. Show formal charges. The skeletal structure of the molecule is C N N
-
(a) Draw two resonance structures of the cation shown below, shifting only one electron pair in each step.- Be sure to include the formal charge on structures B and C. Only move one double bond. Each...
-
The following molecular model is that o a carbocation. Draw two resonance structures for the carbocation, indicating the positions of the doublebonds.
-
Dakota Company had net sales ( at retail ) of $ 1 4 4 , 0 0 0 . \ table [ [ , At Cost,At Retail ] , [ Beginning inventory,$ 3 3 , 3 0 0 , $ 6 5 , 6 0 0 es Dakota Company had net sales (at retail) of...
-
The following transactions occurred between Chans Antique Furniture and M & L Furniture Warehouse during August of the current year: Aug 4 Chans Antique Furniture purchased $6,700 of merchandise from...
-
Use Equation (7.34) (page 545) to derive the formula Equation (7.34) .2 E (x; T)? B1 i=1 ||
-
From 2008 to 2015, auto loan rates in the United States declined from around 8% to near historic lows of around 4%. At the same time, auto sales increased dramatically. How, if at all, does this...
-
Tiger Computers, Inc., of Singapore is considering the purchase of an automated etching machine for use in the production of its circuit boards. The machine would cost $900,000. (All currency amounts...
-
RELATIONAL DATABASE CONCEPTS: Describe the basic steps required to install the Oracle, SQL Server, and MySQL relational database management systems (RDBMSs) and the major challenges that the user may...
-
In some cases, the FriedelCrafts acylation reaction can occur intramolecularly, that is, within the same molecule. Predict the product of the following reaction: CI AICI 3 ?
-
Which of the following compounds are chiral? Label all chirality centers. (a) (d) H3C CH3 | | CH3CHCHCCHCH3 CH3 BrCHCHCHCHBr (b) H3C (e) - H3C 0 CH3 (c) (f) H3C H3C-- H CH3
-
On 30 November 20x1, Systech Ltd entered into a non-cancellable contract to buy 1,000 shares of Fastrack Ltd for $5,000 on 31 July 20x2. On the same date, Systech Ltd purchased a put option on 1,000...
-
X Company has four inventory items at year end. Inventory information (based on FIFO) is as follows: Determine X Companys ending inventory using the lower-of-cost-or-market method. Item 1 2 3 Cost...
-
Emily Eagels owned stock with a basis of $50,000 which she sold to her sister, Janet, for $42,000. Three months later, Janet sold the stock through a broker for $52,000. What is the gain or loss to...
-
On September 16, 2019, Donald Dunn sold for $80,000 property with an adjusted basis of $20,000. The buyer paid $20,000 down with the remaining $60,000 to be paid in four equal annual installments of...
-
Sheryl Sugarman owned 60 percent of the outstanding stock of Octavian Corp. The corporation sold stock (cost $5,000) to her for $2,500. Sheryl sold the stock for $7,000. What is the corporation's...
-
During 2019, Peter Patel sold a piece of land he had purchased for $40,000. The buyer paid cash of $50,000 and transferred to Peter a piece of farm equipment having a fair market value of $30,000....
-
Indicate the effects of the following business transactions on the accounting equation of Vivians Online Video store. Transaction (a) is answered as a guide. a. Received cash of $10,000 from issuance...
-
Halley's comet travels in an ellipti- cal orbit with a = 17.95 and b = 4.44 and passes by Earth roughly every 76 years. Note that each unit represents one astronomical unit, or 93 million miles. The...
-
When ethene gas is passed into an aqueous solution containing bromine and sodium chloride, the products of the reaction are the following: Write mechanisms showing how each product is formed. Br Br-...
-
The double bond of tetrachloroethene is undetectable in the bromine test for unsaturation. Give a plausible explanation for this behavior.
-
Shown below is the final step in a synthesis of an important perfume constituent, cis-jasmone. Which reagents would you choose to carry out this last step? cis-Jasmone
-
During early 2008, the market sentiment and the credit crunch in the United States and worldwide created a lot of pressure on the liquidity of KMTB Finance Company. Even though short-term interest...
-
Your post-secondary institution is facing budget cuts. One of the areas that has been mentioned for a reduction in services is the online writing help (OWH). As a student who has used online writing...
-
for new investments at the firm, At this stage, typically reviewing and negotiating a Confidentiality Agreement with the seller before receiving more detailed information in this stage what are :...

Study smarter with the SolutionInn App


