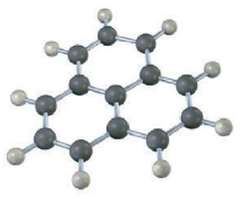
The following molecular model is that o a carbocation. Draw two resonance structures for the carbocation, indicating
Question:
The following molecular model is that o a carbocation. Draw two resonance structures for the carbocation, indicating the positions of the doublebonds.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Three resonance forms for the carbocation of the formula C3H9 ...View the full answer

Answered By

Nidhinkrishna K R
A friendly, approachable and hardworking individual with an exceptional interest in providing tutoring services to assist students in comprehending language concepts. Patient and pleasant, with a demonstrated ability in communicating with people from different backgrounds.
Special talent for:
• Recognizing variations in student backgrounds, abilities and learning styles.
• Interacting with students in a friendly and respectful manner, aiming to comprehend their specific learning abilities and limitations.
• Explaining science in easy to understand ways without overwhelming students.
• Helping students polish their knowledge about science and history , and build upon their personal abilities.
EDUCATION
Pursuing Master’s Degree in Environmental science ,Central University of Kerala.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw two resonance structures for diazomethane, CH2N2. Show formal charges. The skeletal structure of the molecule is C N N
-
The following molecular model is a representation of para-aminobenzoic acid (PABA), the active ingredient in many sunscreens. Indicate the positions of the multiple bonds. And draw a skeletal...
-
(a) Write two resonance structures for the formate ion HCO2-.(The hydrogen and oxygen atoms are bonded to the carbon.) (b) Explain what these structures predict for the carbon-oxygen bond lengths of...
-
The B.B. Lean Co. has 1.4 million shares of stock outstanding. The stock currently sells for $20 per share. The firm's debt is publicly traded and was recently quoted at 93 percent of face value. It...
-
Suppose that when Reed was seeking to hire a certified financial planner, he asked you for advice on where to recruit this person. Which sources would you suggest, and why?
-
Rhodium has an atomic radius of 0.1345 nm and a density of 12.41 g/cm3. Determine whether it has an FCC or BCC crystal structure.
-
Robert Boyle (1627-1691) established the law that (pressure \(\times\) volume) \(=\) constant for a gas at a constant temperature. By pouring mercury into the open top of the long side of a...
-
Hamilton Hawks Players' Association and Mr. Sideline, the CEO and majority owner of Hamilton Hawks Soccer, Inc, ask your help in resolving a salary dispute. Mr. Sideline presents the following income...
-
72 Question 1 (50 marks) a) Identify all zero-force members in the truss. [10 marks] b) Using the method of sections, compute the forces in the members BC, BF, EI and EF (identified by x in the...
-
Dominique Fouque owns and operates Dominique's Doll House. She has a small shop in which she sells new and antique dolls. She is particularly well known for her collection of antique Ken and Barbie...
-
A 1, 6-Methanonaphthalene has an interesting 1 H NMR spectrum in which the eight hydrogen?s around the perimeter absorb at 6.9 to 7.3 ?, while the two CH 2 protons absorb at ?0.5 ?. ?Tell whether it...
-
Azulene, an isomer of naphthalene, has a remarkably large dipole moment for a hydrocarbon (? = 1.0 D). Explain, using resonance structures. Azulene
-
Table 11.10 refers to a three-period crossover trial to compare placebo (treatment A) with a low-dose analgesic (treatment B) and high-dose analgesic (treatment C) for relief of primary dysmenorrhea....
-
For each quadratic relation given in standard form, determine key features (x-intercepts, y-intercept, equation of the axis of symmetry and the coordinates of the vertex) of the graph using both the...
-
In table format , lay out the following stats for the most recent 5 years on the company (include the correct symbol as well, e.g. %, x, days, etc). Below the chart in a few sentences explain the key...
-
3. Find (1) the time ratio and (2) length of stroke of the following quick-return mechanism where ao bo-20, ao a=40, bo b=25, bc=100. Pw \/b bo PR 5 Lofo- S 6
-
Using the z-scores table for the standard Normal distribution In finance, the acronym VaR stands for "value at risk". JP Morgan introduced value at risk in the 1980s as a way to answer a common...
-
You are looking to calculate a price per share on Troy Inc. You have estimated the following future free cash flows. The WACC is 7%, there are 15 million shares outstanding, the MV of debt and...
-
True of False: Ifmoneyisworth6%compoundedannually to you, then you should prefer toreceive\($1750\)peryearfor5yearsthantoreceive\($1000\)peryear for10years,assuming the first receipt occurs one year...
-
How will relating product contribution margin s to the amount of the constrained resource they consume help a company maximize its profits?
-
Explain the difference between K c and K p . For a given reaction, how are the two constants related?
-
Predict the products obtained when D-galactose reacts with each reagent. (a) Br2 and H2O (b) NaOH, H2O (c) CH3OH, H+ (d) Ag(NH3)2+-OH (e) H2, Ni (f) Excess AC2O and pyridine (g) Excess CH3I, Ag2O (h)...
-
Draw the following sugar derivatives. (a) Methyl -D-glucopyranoside (b) 2, 3, 4, 6-tetra-O-methyl-D-mannopyranose (c) 1, 3, 6-tri-O-methyl-D-fructofuranose (d) Methyl 2, 3, 4,...
-
Draw Fischer projections for the enantiomers of threo-hexane-1, 2, 3-triol. HOCH2 - CH(OH) - CH(OH) - CH2CH2CH3
-
Skysong needs to calculate the company's expected cash receipts for the upcoming month to determine whether additional financing is needed. Typically, the company's sales consist of 40% cash sales...
-
The value of NSE index is 1140. The continously compunded risk free rate is 4.6% amnd continous dividend yield is 2.1%. Calculate the no arbitrage price for a 140 day forward contract on the index
-
Explain why MNCs such as Coca Cola and PepsiCo, Inc., still have numerous opportunities for international expansion. Explain briefly

Study smarter with the SolutionInn App


