Use Figure 1.14 to order the following molecules according to increasing positive character of the carbon atom:
Question:
Use Figure 1.14 to order the following molecules according to increasing positive character of the carbon atom:![]()
Fig 1.14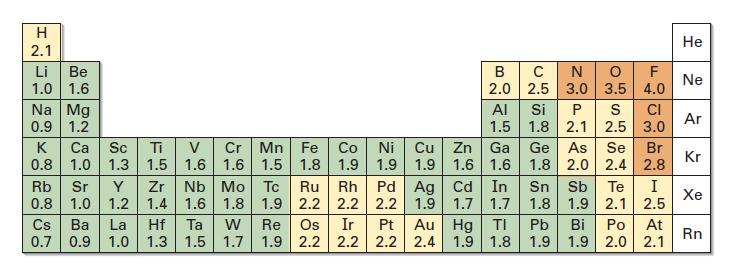
Transcribed Image Text:
CH3F, CH₂OH, CH3Li, CH3I, CH3CH3, CH3NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To determine the increasing positive character of the carbon atom in the given molecules we can refe...View the full answer

Answered By

Churchil Mino
I have been a tutor for 2 years and have experience working with students of all ages and abilities. I am comfortable working with students one-on-one or in small groups, and am able to adapt my teaching style to meet the needs of each individual. I am patient and supportive, and my goal is to help my students succeed.
I have a strong background in math and science, and have tutored students in these subjects at all levels, from elementary school to college. I have also helped students prepare for standardized tests such as the SAT and ACT. In addition to academic tutoring, I have also worked as a swim coach and a camp counselor, and have experience working with children with special needs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Name the following molecules according to the IUPAC system of nomenclature. (a) (b) (c) (d) (e) CH3CH(CH3)CH(CH3)CH(CH3)CH(CH3)2 (f) CH3CH2CHCH3 CH CH H3C CH3 CHCHCH2CH3 EH CHCH,CH.CCH,CH.CH CH,...
-
Name the following molecules according to the IUPAC nomenclature system. (a) (b) (c) (d) (e) (f) CH32 Cl CH3 Br
-
On September 30 of the current year, Silver Fox Corporation files for bankruptcy. At the time, it estimates that the total FMV of its assets is $725,000, whereas the total amount of its outstanding...
-
Under what circumstances may an involuntary petition for relief be filed? Who files this petition?
-
What types of jobs will probably remain plentiful in the future, and how can you prepare for them?
-
Shiloh supplies equipment to the automotive and commercial vehicle markets and other industrial customers. It specializes in materials and designs that reduce vehicle weight and increase fuel...
-
Canon, a Japanese manufacturer of photographic equipment, decided against offshoring and kept its manufacturing and new product development processes in Japan, which has relatively high labor costs,...
-
1. Solve the double inequality below. Then graph the solution set on the real number line. -81+3(x-2) < 13
-
How many valence electrons does each of the following atoms have? (a) Oxygen (b) Magnesium (c) Fluorine
-
The following model is that of aspartame, C 14 H 18 N 2 O 5 , known commercially under many names, including NutraSweet. Only the connections between atoms are shown; multiple bonds are not...
-
The active ingredients in a particular antacid tablet are aluminum hydroxide, Al(OH) 3 , and magnesium hydroxide, Mg(OH) 2 . A 5.00 x 10 2 mg sample of the active ingredients was dissolved in 50.0 mL...
-
Kleslia, Inc. budgeted manufacturing overhead of $365,224 at a level of 9,764 direct labor hours. The actual results at the end of February showed 2,338 direct labor hours and total manufacturing...
-
In Orality and Literacy, Walter Ong explains how the evanescence of speech, coupled with the limitations of human memory, produce for "primary oral societies" - societies with neither writing nor...
-
Feldman argues that members of a discourse community have a responsibility to use the community's lexis (para. 6). Why does she say so? Does it seem strange to you to imagine a writer having a...
-
1) search social media sites to get an idea of how the public may perceive the company? 2) learn what you can about the senior executives of the company and their plans?
-
When the velocity v of an object is very large, the magnitude of the force due to air resistance is proportional to v with the force acting in opposition to the motion of the object. A shell of mass...
-
On average, 2.4 customers per minute arrive at an airline check-in desk during the peak period. Assume that the distribution of arrivals is Poisson. a. What is the probability that there will be no...
-
What is a make-or-buy decision?
-
The organoborane used in a Suzuki reaction is prepared by the reaction of catecholborane with an alkene or an alkyne. What hydrocarbon would you use to prepare the organoborane of Problem 35? Problem...
-
The mass spectra of two very stable cycloalkanes both show a molecular ion peak at m/z = 98. One spectrum shows a base peak at m/z = 69, the other shows a base peak at m/z = 83. Identify the...
-
Which of the following compounds has a vibration that is infrared inactive: acetone, 1-butyne, 2-butyne, H 2 , H 2 O, Cl 2 , ethene?
-
Find the amount of work and heat exchanged for two moles of ideal monatomic gas undergoing the process shown in the figure. P, kPa 120 80.0 10.0 16.0 V, L
-
Paraphrase and rewrite in own words: Defending your network is easier if you have the right equipment and software in place. Preventing rogue access points and other unauthorized devices from joining...
-
. A rock is kicked horizontally off a 321 m tall bridge. The rock hits the water so that the horizontal displacement is 45.0 m. What was the initial velocity the rock was kicked?

Study smarter with the SolutionInn App


