Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that
Question:
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do.
a. CHF=CHF
b. F2C=CH2
c. CH2=CH-CH2-CH3
d.
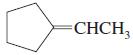
e.
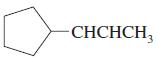
f.
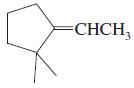
Transcribed Image Text:
CHCH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
The cis trans isomerism is shown by the alkenes only The essenti...View the full answer

Answered By

Mr. VISHAL
I'm pursuing msc. Chemistry (organic specialisation). I have deep knowledge of chemistry and i can explain the concepts in a very simple way. I also like to give lectures on specific topics.
My linkedin profile is:
www.linkedin.com/in/vishal-chnaliya-66a85514b
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may...
-
Which of the following compounds show only a single peak in their 1H NMR spectrum? a. CH3CH2OCH2CH3 b. c. CH,CH,CCI
-
Which of the following compounds are aromatic? a. b. c. Cycloheptatrienyl cation d. e. f. g. Cyclononatetraenyl anion h. CH2=CHCH=CHCH=CH2
-
In Exercises find the positive values of p for which the series converges. n=1 n
-
BH+ ClO-4 is a salt formed from the base B (Kb = 1.00 10-4) and perchloric acid. It dissociates into BH+, a weak acid, and ClO-4, which is neither an acid nor a base. Find the pH of 0.100 M BH+ClO-4.
-
Calculate the total work, in Btu, produced by the process of Fig. P45E. 500 100 O. 2 4 V, ft3 P, psia
-
What is the rate of return for a $\$ 14,000,20$-year investment that provides a payout of $\$ 1,000$ a year?
-
Brandi just received her credit card bill, which has an outstanding balance equal to $3,310. After reviewing her financial position, Brandi has concluded that she cannot pay the outstanding balance...
-
Melissa recently paid $660 for round-trip airfare to San Francisco to attend a business conference for three days. Melissa also paid the following expenses: $650 fee to register for the conference,...
-
Following are financial statements for Moore Company and Kirby Company for 2021: Moore purchased 90 percent of Kirby on January 1, 2020, for $657,000 in cash. On that date, the 10 percent...
-
Two compounds with the formula CH 3 -CH=N-CH 3 are known. (a) Draw a Lewis structure for this molecule, and label the hybridization of each carbon and nitrogen atom. (b) What two compounds have this...
-
Give the relationship between the following pairs of structures. The possible relationships are: same compound cis-trans isomers constitutional isomers (structural isomers) not isomers (different...
-
The way in which planning and control are integrated within the business cycle is called the ______-______ ______.
-
A dysfunctional group of hopeful, unpublished writers attempts to remain unconditionally positive and supportive in their communication in the face of constant rejection. Not for everyone, but this...
-
Define working capital.
-
Ltd processes and purifies a basic chemical which is then broken down by reaction to give three separate products. Explain the approaches to joint cost allocation using the following information:...
-
What have researchers found about the length of the working capital cycle in practice?
-
Is hypercompetitive communication ethical? Explain.
-
Using the data presented in BE16-5, assume Strand Corp. is a public company and that it purchased Hydrocor's bonds to trade. Prepare the journal entries to record (a) The purchase of the bonds on...
-
Find the center of mass of a thin triangular plate bounded by the y-axis and the lines y = x and y = 2 - x if (x, y) = 6x + 3y + 3.
-
What products are formed when gach of the following ethers reacts with concenffated aqueous HI? 2-ethoxy-2,3-dimethylbutane
-
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.) C,H OH/H,O CH CH2 sodium azide
-
Suggest a Williamron other cynthosis, if one is possible, for each of the following compounds. If no Williamson ether synthesis is possible, explain why. (CH3)2CH---S---CH3
-
Solve (2.4 x 10-5) X 875 (2.5 x 107) x (2.8 x 107)
-
assume there are no outliers. Draw the boxplot for the following frequency table. You may X f rf cf crf 3 7 0.1228 7 0.1228 4 11 0.1930 18 0.3158 5 9 0.1579 27 0.4737 618 6 0.1053 33 0.5789 10 0.1754...
-
Find the partial derivative of the regularized least squares problem: {) (w + x) (wo + w x ( ) + w x ( ) } + |/\||[w1, w2]|| with respect to wo, w, and w2. Although there is a closed-form solution to...

Study smarter with the SolutionInn App


