A box labeled C 6 H 10 isomers contains samples of three compounds: A, B, and C.
Question:
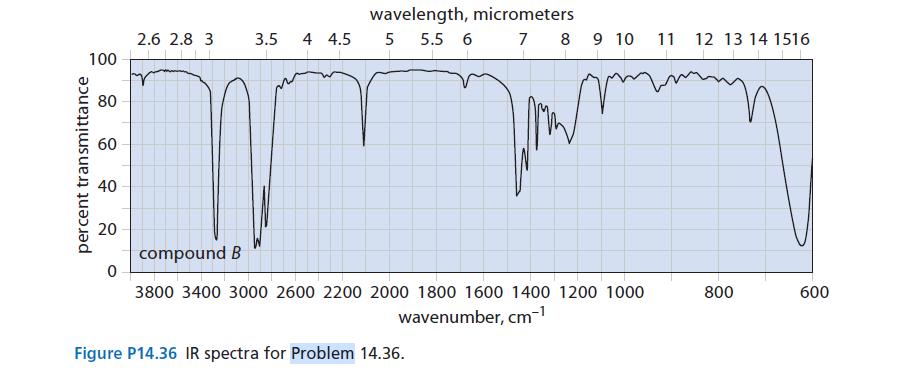
A box labeled “C6H10 isomers” contains samples of three compounds: A, B, and C. Along with the compounds are the IR spectra of A and B, shown in Fig. P14.36. Fragmentary data in a laboratory notebook suggest that the compounds are 1-hexyne, 2-hexyne, and 3-methyl-1,4-pentadiene. Identify the three compounds.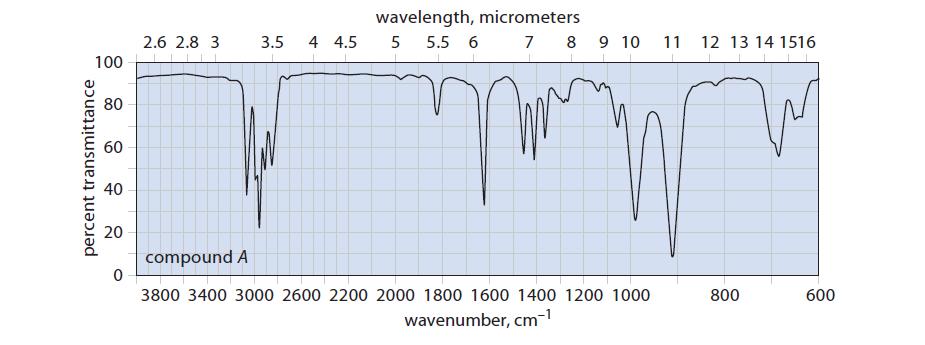
Transcribed Image Text:
percent transmittance 100 80 60 40 20 0 2.6 2.8 3 3.5 4 4.5 wavelength, micrometers 5 5.5 6 7 8 9 10 11 12 13 14 1516 m compound A 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm-¹ 800 600
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
First look for the telltale CH and CC stretching absorpt...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The IR and mass spectra for three different compounds are shown in Figures 13.45-13.47. Identify each compound. a. b. c. 25 26 2.7 28 19 2000 100 43 58 100 71 85 0 20 40 60 80 100 120 m/z 13 14 15 6...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. The Incisors own a rental beach house in Hawaii. The beach house was rented for the full year during 2012...
-
There are several problems that are unique to family businesses or ventures. Which of the following is not one of those problems? Issues of succession Problems of control, fairness, and equity Issues...
-
Carters Produce has applied for a loan and has agreed to use its inventory to collateralize the loan. The company currently has an inventory balance of $289,000. The cost of goods sold for the past...
-
The comparative balance sheet of Jackson Educational Supply at December 31, 2015, reported the following: Jacksons transactions during 2015 included the following: Requirements 1. Prepare the...
-
Why has the importance of environmental accounting to business grown in the past ten years?
-
Allowance Method of Accounting for Bad DebtsComparison of the Two Approaches Kandel Company had the following data available for 2010 (before making any adjustments): Accounts receivable, 12/31/10...
-
Dog Up ! Franks is looking at a new sausage system with an installed cost of $ 6 7 0 , 0 0 0 . This cost will be depreciated straight - line to zero over the project s 5 - year life, at the end of...
-
(a) Draw the structures of all enols that would spontaneously form the following ketone, including stereoisomers. (b) Would alkyne hydration be a good preparative method for this compound? If so,...
-
Give the principal product(s) expected when 1-hexyne or the other compounds indicated are treated with each of the following reagents: (a) HBr (b) H, Pd/C (c) H, Pd/C, Lindlar catalyst (d) product of...
-
True or false? A theory is an attempt to explain why a law is true.
-
The equation of motion of a particle travelling along the \(x\)-axis is given as where \(b=0.4 \mathrm{~m} / \mathrm{s}^{3}, c=0.5 \mathrm{~m} / \mathrm{s}^{2}\) and \(d=50 \mathrm{~m} /...
-
When a certain ideal gas thermometer is immersed in a water-ice mixture at \(273.15 \mathrm{~K}\), the mercury level in the right arm is \(102 \mathrm{~mm}\) above the reference mark. When this...
-
Create another class file called PrintTest2 and save it as a .java file. Create a main() container inside PrintTest2. Inside the main() container, write code that will print out the following on the...
-
Assume that the MapReduce job from Page #438 in Section 6.2 is executing a task with 2^40 bytes of input data, 2^37 bytes of intermediate data, and 2^30 bytes of output data. This job is entirely...
-
Nkululeko J. Ntshanga owns a manganese ore mining business in South Africa. He is interested in attracting additional investors to obtain financing for planned expansion. Some potential investors...
-
The following data for Kitchen Tile Company relates to the production of 18,000 tiles during the past month. The company allocates fixed overhead costs at a standard rate of $19.00 per direct labor...
-
Danielle has an insurance policy with a premium of $75 per month. In September she is in an accident and receives a bill worth $2990 for the repair of her own property. Her deductible is $250 and her...
-
Would you expect a conjugated diyne such as 1, 3-butadlyne to undergo DielsAlder reaction with a dienophile explain.
-
Reaction of isoprene (2-methyl-1, 3-hutadiene) with ethyl propenoate gives a mixture of two Diels?Alder adducts. Show the structure of each, and explain why a mixture is formed. CO2CH2CH3
-
Rank the following dienophiles in order of their expected reactivity in the Diels?Alder reaction. CH CH NC NC CN c=C CH c=c c=C c=C NC CN
-
3. ABC has the following data: (in thousands) Louisian a Mississippi Other Total Sales 600 300 100 1000 Payroll 80 40 120 Propert y 200 400 600 Their federal taxable income for the year is $500,000....
-
During 2024, its first year of operations, Laminate Construction provides services on account of $144,000. By the end of 2024, cash collections on these accounts total $102,000. Laminate estimates...
-
2. After scouting a number of locations, your financial analysts have settled on a desirable location for one of the firm's new manufacturing plants. However, the state income and local property...

Study smarter with the SolutionInn App


