(a) Which of the compounds shown in Fig. P6.40 can in principle be resolved into enantiomers? Explain...
Question:
(a) Which of the compounds shown in Fig. P6.40 can in principle be resolved into enantiomers? Explain why or why not.
(b) Omeprazole can be separated into two enantiomers that do not interconvert at room temperature.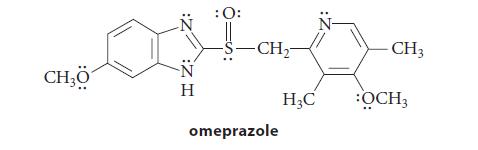
(c) Esomeprazole, the S enantiomer of omeprazole, is a drug used to control acid reflux. Redraw the structure of omeprazole in part (b) to show it as the S enantiomer.
Transcribed Image Text:
CH₂0 :Z H Η : 0: -S-CH₂- omeprazole H₂C - CH3 :OCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Any compound that is chiral can in principle be resolved into enantiomers Compound A possesses a p...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds could in principle be resolved into enantiomers at very low temperatures? Explain. (a) Propane (b) 2,2,3,3, -tetramethylbutane
-
Which of the following compounds could be resolved into enantiomers at room temperature? Explain. (a) (b) CH2CH CH3)2 CH CH CH3 CH3
-
Glorious Electrical Appliances (GEP) Co. is a company that sells electrical tools. GEP uses perpetual inventory system in recording its inventory. The financial position of GEP as at 31 December 2016...
-
The Ramirez Company current dividend was $1.90. Its dividend will grow by 6%, 7%, 8% and 9% for the first 4 years. And then, dividends to grow at a rate of 5% forever. It's required return is 15%. 1-...
-
Make an argument that what McElveen and friends did indicates nothing immoral about them but rather something wrong with society.
-
Petro Corporation is a U.S. corporation with operations in several foreign countries as well as in the United States. During the current year, Petros worldwide taxable income is $600,000. Petros...
-
Two conspirators, Timothy Carpenter and Timothy Sanders, were charged with aiding and abetting a robbery affecting interstate commerce and the use or carriage of a firearm. At trial, the FBI...
-
Activity-based costing, service company Quikprint Corporation owns a small printing press that prints leaflets, brochures, and advertising materials. Quikprint classifies its various printing jobs as...
-
Commencing 1 April 2022, Morgan Landscaping Pty Ltd (morgan) provided Larissa (an employee) with the use of a Volkswagen Golf car. The car cost $55,000 (including GST) and was used privately every...
-
The specific rotation of the R enantiomer of the following alkene is [a] 25 D 5 179 degrees mL g 1 dm 1 , and its molecular mass is 146.2. (a) What is the observed rotation of a 0.5 M solution of...
-
Construct sawhorse and Newman projections (Sec. 2.3A) of the three staggered conformations of 2-methylbutane (isopentane) that result from rotation about the C2C3 bond. (a) Identify the conformations...
-
Two long straight thin wires with current lie against an equally long plastic cylinder, at radius R = 20.0 cm from the cylinder's central axis. Figure ??a?? shows, in cross section, the cylinder and...
-
You will play the role of a customer in a restaurant. Choose a restaurant that you have never eaten at before and that has wait-staff to serve you at your table ( not a self-serve, fast food or...
-
Critically explain the political factors affecting Air canada. please give detailed explanation and also add references
-
1. Identify two firms in two different industries that you believe exhibit a nimble nature in their operations. 2. What specific evidence leads you to believe these firms are nimble, especially in...
-
Project selection sets an organization on the path to use of its limited resources. Without a sound project selection method, an organization may not be as efficient as they otherwise could be....
-
December 31, 2030 Accounts Debit Credit Cash Accounts Receivable 105,800 14,600 Allowance for Bad Debts Inventory Prepaid Advertising Supplies Accounts Payable Unearned Revenue Long-Term Note Payable...
-
Which of the following are short-run and which are long-run adjustments? a. Wendys builds a new restaurant. b. Harley-Davidson Corporation hires 200 more production workers. c. A farmer increases the...
-
Find the area of the surface generated by revolving the para- metric curve x = cos 1, y = sin? 1 (0 < I sa/2) about the y-axis.
-
Predict the major product(s) of nitration of the following substances. Which react faster than benzene and which slower? (a) Bromobenzene (b) Benzonitrile (c) Benzoic acid (d) Nitrobenzene (e)...
-
Rank the compounds in each group according to their reactivity toward electrophilic substitution. (a) Chlorobenzene, o-dichlorobenzene, benzene (b) p-Bromonitrobenzene, nitrobenzene, phenol (c)...
-
A Predict the major mono-alkylation products you would expect to obtain from reaction of the following substances with chloromethane arid AlCl3: (a) Bromobenzene (b) m-Bromo-phenol (c)...
-
1. Write the class declaration of Manager. < > Executive Manager Employee 2. Draw the UML diagram from the class declaration below. public class Engineer extends Employee implements Serializable,...
-
8. (Expected rate of return and risk) Syntex, Inc. is considering an investment in one of two common stocks. Given the information that follows, which investment is better, based on the risk (as...
-
Let S be the set of binary strings consisting of a (nonempty) block of Os followed by a (nonempty) block of 1s, such that if the block of Os has odd length, then the block of 1s has even length. Let...

Study smarter with the SolutionInn App


