Complete the reactions given in Fig. P27.70 assuming the amino acid residue is part of a peptide
Question:
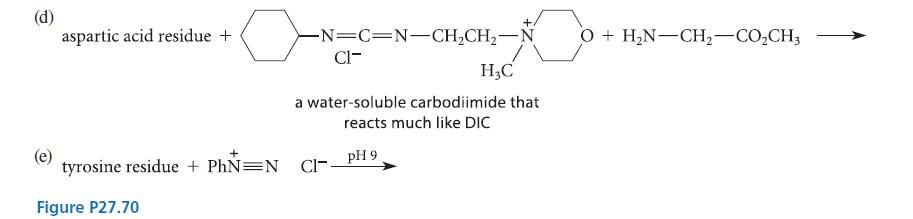
Complete the reactions given in Fig. P27.70 assuming the amino acid residue is part of a peptide in aqueous solution and is at neither the amino nor the carboxy terminus.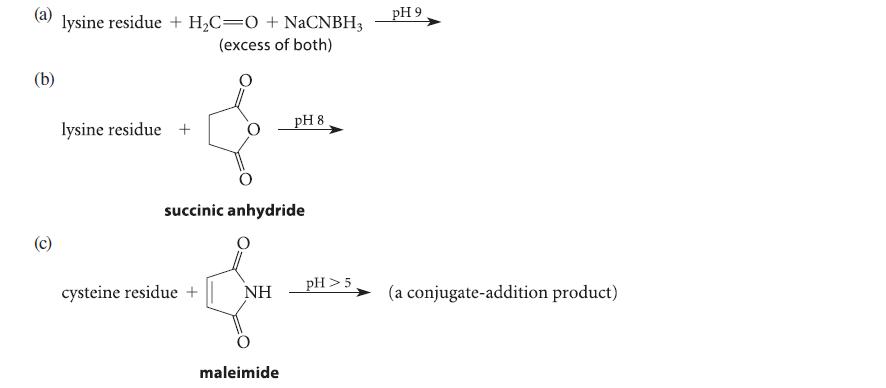
Transcribed Image Text:
(a) lysine residue + H₂C=O + NaCNBH3 (excess of both) (b) (c) lysine residue + succinic anhydride cysteine residue + NH pH 8 maleimide pH > 5 pH 9 (a conjugate-addition product)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a b This is a reductive amination of formaldehyde by the amino group of the lysine residue Because e...View the full answer

Answered By

Niala Orodi
I am a competent and an experienced writer with impeccable research and analytical skills. I am capable of producing quality content promptly. My core specialty includes health and medical sciences, but I can competently handle a vast majority of disciplines.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete the reactions given in Fig. P26.61, assuming the amino acid residue is part of a peptide in aqueous solution and is at neither the amino nor the carboxy terminus. (a) (b) plI 8 lysine...
-
Complete the reactions given in Fig. P19.45 by giving the principal organic product(s). Fig. P19.45 (a) (b) (c) p-toluenesulfonic acid (catalyst CH, t CH,OH - (solvent) ether Hio -caphenone, +...
-
Complete the reactions given in Fig. P22.81 by giving the major organic products. Explain your reasoning. NaOEt excess) EtOH H,o heat CHsI (c) CI NO, H ot, heat C CH LiAIH 2) HaO (e) CH Et CHCHCOEOH...
-
Alert Security Services Co. offers security services to business clients. The trial balance for Alert Security Services Co. has been prepared on the following end-of-period spreadsheet for the year...
-
(Multiple Choice Question) 1. Miga Company and Porter Company both bought a new delivery truck on January 1, 2008. Both companies paid exactly the same cost, $30,000, for their respective vehicles....
-
Lawrence Products had a beginning balance in Furniture and Fixtures, net of depreciation, of $ 192,000. The ending balance in that account was $ 196,500. Depreciation expense for that period was $...
-
A university pumps its water from wells located on campus. The falling water table has caused pumping costs to increase, the quantity of water available to decrease, and the quality of water to...
-
The following transactions were completed by The Spencer Gallery during the current fiscal year ended December 31: Mar. 15. Reinstated the account of Brad Atwell, which had been written off in the...
-
Let M, N be closed subspaces of a Hilbert space H and P, Q the orthogonal projections with ran P = M, ran Q = N. Prove that the following conditions are equivalent:
-
Poly-l-lysine (a peptide containing only lysine residues) exists entirely in an a-helical conformation at pH > 11. Below pH 10, however, the peptide becomes a random coil. Poly-l-glutamic acid, on...
-
Complete the reactions given in Fig. P27.75 by giving the structure of the major organic product(s). (a) ethylamine + Ph-N-C=S (b) (d) PhCH=O + KCN + CH3NH, NaOH (1 equiv.) + HNCH-CO; I CH3 CH3...
-
A local brewery wishes to ensure that an average of 12 ounces of beer is used to fill each bottle. In order to analyze the accuracy of the bottling process, the bottler takes a random sample of 48...
-
Assume the CAPM holds. Investors are only allowed to invest in stock A and stock B (these two securities form the market portfolio). Each asset's weight in the market portfolio is simply its weight...
-
Acrobat you Brighton, Inc. is currently a C corporation. Brighton has 10 shareholders and has substantial amounts in retained earnings from previous years. Brighton's officers are considering...
-
Conceptually, NP represents the class of decision problems whose "yes" instances can be efficiently verified (without incorrectly accepting any "no" instances). In this problem, you will consider its...
-
The Rocky Mountain Resort Corporation owns and operates a luxury hotel in Banff, Alberta. In response to complaints about patrons bringing their dogs and cats into the hotel, the resort has...
-
Suppose in February, a palm oil producer anticipates that he will have 180 metric tonnes of crude palm oil ready for sale in four months' time. He would like to fix the price for his produce. The...
-
The Johnson County Arts Foundation aims to promote the appreciation and practice of art in Johnson County and surrounding areas. Consistent with this mission, the foundation sponsors a number of...
-
Smthe Co. makes furniture. The following data are taken from its production plans for the year. Required: 1. Determine the hazardous waste disposal cost per unit for chairs and for tables if costs...
-
Which of the following alkenes can exist as double-bond stereo? Identify the stereo centers in each. (a) CH3CH2CH=====CHCH2CH3 (b) CH,CIH CH-CCH CH
-
Which of the following cannot be, correct formula (r) for an organic compound? Explain (a) C10H20N3 (b)C30H20N2O2 (c) C10H27N3O2 (d) C10H16O;
-
Which compound in each set should have the larger dipole moment? Explain. Propene or 2-methylpropene
-
What do you see as the United States' biggest domestic terrorism threat today
-
The recent pandemic crisis followed by the war in Ukraine has halted the process of globalization. What are some of the effects of a globalization contraction?
-
What industry sectors are most likely to add jobs during a down economy? During an improving economy? Are there employment sectors that are essentially unaffected by economic fluctuations?

Study smarter with the SolutionInn App


