Complete the reactions given in Fig. P26.61, assuming the amino acid residue is part of a peptide
Question:
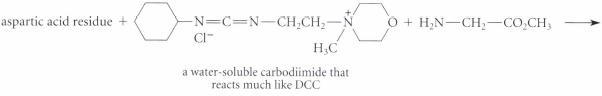
Complete the reactions given in Fig. P26.61, assuming the amino acid residue is part of a peptide in aqueous solution and is at neither the amino nor the carboxy terminus.
(a)
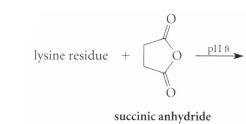
(b)
Transcribed Image Text:
plI 8 lysine residue + succinic anhydride
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
a The sidechain amino group of lysine serves as a nucleophile to ...View the full answer

Answered By

Shadrack Mulunga
I am a Biochemistry by profession. However, I have explored different fields of study. My quest to explore new fields has helped me gain new knowledge and skills in Business, clinical psychology, sociology, organizational behavior and general management, and Project Management. I count my expertise in Project management, in particular, creation of Work Break Down Structure (WBS) and use of Microsoft Project software as one of my greatest achievement in Freelancing industry. I have helped thousands of BSC and MSC students to complete their projects on time and cost-effectively using the MS Project tool. Generally, I find happiness in translating my knowledge and expertise to success of my clients. So far, i have helped thousands of students to not only complete their projects in time but also receive high grades in their respective courses. Quality and timely delivery are the two key aspects that define my work. All those who hired my services always come back for my service. If you hire my services today, you will surely return for more. Try me today!
5.00+
154+ Reviews
289+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using known reactions and mechanisms discussed in the text, complete the reactions given in Fig. P19.46 on p. 942. Fig. P19.46 NaBH4 CH OH
-
Complete the reactions given in Fig. P21.52 by giving the principal organic products. Explain how you arrived at your answers. NaOH CH O (trace) H,C CCHO CH +CH OH (solvent) Ph NH2 1 (CgH,NO3)...
-
Complete the reactions given in Fig. P22.81 by giving the major organic products. Explain your reasoning. NaOEt excess) EtOH H,o heat CHsI (c) CI NO, H ot, heat C CH LiAIH 2) HaO (e) CH Et CHCHCOEOH...
-
As programs become more complex, it becomes increasingly important to plan or "design" your code before writing it. Designing your code will help you organize its logic. It will also help you keep...
-
Predator Pucks, Inc. has current assets of $8,000, net fixed assets of $45,000, current liabilities of $6,800, and long-term debt of $13,800. What is the value of the shareholders' equity account for...
-
A man pushing a mop across a floor causes it to undergo two displacements. The first has a magnitude of 150 cm and makes an angle of 120 with the positive x axis. The resultant displacement has a...
-
A study on factors that may be related to the number of weeks a person has been jobless has been conducted in Cheyenne and Memphis. The data collected in Cheyenne are provided in the file...
-
Misty Higgin, manager of the Hass Music Hall, is considering the opportunity to expand the companys concession revenues. Specifically, she is considering whether to install a popcorn machine. Based...
-
Two balls, one with a mass of 2 kg and the other with a mass of 3 kg, collide elastically. After the collision, the 2-kg ball moves to the left with a velocity of 4 m/s and the 3-kg ball moves to the...
-
Determine the monthly cash flows and total cash generated at the end of each month and just before the payment is received for the construction of a house with the following budget and schedule. On...
-
Draw the structure of the major neutral form of each of the following peptides, G-D-G-L-F
-
Show how the acetamidomalonate method can be used to prepare the following unusual amino acids from the indicated starting material and any other reagents. (a) (b) (c) (CH) CDCH CH CO from...
-
Preparing a trial balance Required On December 31, 2013, Magee Company had the following normal account balances in its general ledger. Use this information to prepare a trial balance. Common Stock...
-
Tom is a director of Marketing at XYZ Inc. Three years ago, he hired Jane as his secretary. One day, Jane asked Tom over to her house for dinner. They spent the night together, having a pleasant...
-
Why is it important that the characteristics of our population of interest be adequately identified when designing our sample?
-
6. Three identical small Styrofoam balls (m 1.83g) are suspended from a fixed point by three nonconducting threads, each with a length of 50.8cm and with negligible mass. At equilibrium the three...
-
The nucleus of a certain atom is 6.00 fm in diameter and has 109 protons. What is the acceleration of another proton 9.60 fm from the surface of the nucleus? O 8.68x1026 m/s2, away from the nucleus O...
-
Problem 3: Dorado and Maya are the children of Jess and Sabel. In November 2000 Sabel died intestate leaving P2,000,000 estate before P1,000,000 deductions and P15,000 estate tax. How would the...
-
What is a collective bargaining agreement?
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity...
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
Propose a structure for a compound C15H24O that has the following 1H NMR spectrum. The peak marked by an asterisk disappears when D2O is added to thesample. TMS 10 8. 0 ppm Chemical shift (8)...
-
What is Fibonacci heap? Explain CONSOLIDATE operation with suitable example for Fibonacci heap ?
-
Discuss the impact of global supply chain disruptions, exacerbated by events like the COVID-19 pandemic, on inventory management strategies and market resilience across various industries?
-
A pharmaceutical retailer decided to host a website for home delivery of medicines according to user orders. The web application is deployed on a single Amazon EC2 instances. within a few months, the...

Study smarter with the SolutionInn App


