Determine the isomeric relationship between the following two molecules: CH3CH H CHCH3 H H C=C CH3CH CHCH3
Question:
Determine the isomeric relationship between the following two molecules: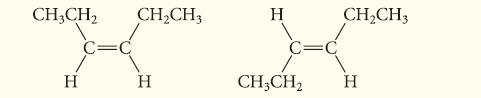
Transcribed Image Text:
CH3CH₂ H CH₂CH3 H H C=C CH3CH₂ CH₂CH3 H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Work from the top of Fig 611 and answer each question in turn These two molecules ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The two isomeric compounds below are naturally occurring insect pheromones. The isomer on the left attracts the male olive fruit fl y; the one on the right, the female. (a) What kind of isomeric...
-
Get-the-lead-out manufactures generic #2 pencils. Its cost function is C(q) 1250 +2q, where q is the number of boxes of pencils it produces each quarter (throughout, quantities and prices refer to...
-
Your team is faced with a puzzle. Four isomeric compounds, A D, with the molecular formula C 4 H 9 BrO react with KOH to produce E G with the molecular formula C 4 H 8 O. Molecules A and B yield...
-
0 The initial substitution of x = a yields the form Simplify the function algebraically, or use a table or 0 graph to determine the limit. When necessary, state "DNE". x + 4x-5 x - 1 Step 1: Factor...
-
Why did the judge direct a verdict in favor of Armani?
-
Sosna Company has a department that manufactures wood trusses (wood frames used in the construction industry). The following information is for the production of these trusses for the month of...
-
Many researchers are interested in the transcription of protein-encoding genes in eukaryotes. Such researchers want to study mRNA. One method that is used to isolate mRNA is column chromatography....
-
Depreciation for Partial PeriodSL, SYD, and DDB Alladin Company purchased Machine #201 on May 1, 2010. The following information relating to Machine #201 was gathered at the end of May....
-
1. Two-point boundary value problem with Dirichlet condition. Consider the following second order elliptic boundary value problem in the one-dimensional space: - -u" (x) = 2, x = (0, 1), u(0) = 0,...
-
Show the planes and centers of symmetry (if any) in each of the following achiral objects. (a) The methane molecule (b) A cone (c) The ethylene molecule (d) The trans-2-butene molecule (e) The...
-
A sample of (S)-2-butanol has an observed rotation of 12.18 at 20 C. The measurement was made with a 2.0 M solution of (S)-2-butanol in methanol solvent in a sample container that is 10 cm long. What...
-
The quotient of 19 and a number d increased by seven. Which of the following is an expression for the statement above? 19 A) +7 d B) C) D) d 19 +7 19+d 7 d+7 19
-
Integrate real-world examples of how social media presence in law enforcement organizations was helpful to the agency. In other words, what are the positive aspects of law enforcement using social...
-
In today's generation, social media has a large impact on society. Discuss the three factors that should be considered during the process of social media management. Secondly, do you believe law...
-
Why is growing the number of users such an important metric for social media companies? How does Metcalfe's Law relate to the profitability of social media companies?
-
There is much misinformation about COVID vaccines being disseminated on Facebook and other social media. Assume Congress passed a law prohibiting Facebook and other social media platforms from...
-
In a common law legal system, only governments can create a Statutory Law. In relation to the media, what would be a media policy? Or What is the major difference between a policy and a law with...
-
The income elasticities of demand for movies, dental services, and clothing have been estimated to be + 3.4, + 1, and + .5, respectively. Interpret these coefficients. What does it mean if an income...
-
The following selected information was taken from Sun Valley Citys general fund statement of revenues, expenditures, and changes in fund balance for the year ended December 31, 2019: Revenues:...
-
Tert-Butyl ethers can be prepared by the reaction of an alcohol with 2-methyipropene in the presence of an acid catalyst. Propose a mechanism for this reaction.
-
Meerwein?s reagent, triethyloxonium tetra-fluoroborate, is a powerful ethylating agent that converts alcohols into ethyl ethers at neutral pH. Show the reaction of Meerwein?s reagent with...
-
Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol). CH2CH=CH2 Safrole
-
Calculate residual income model of dmci for 10 years? Y1 Y2 Y3 Y4 Y5 Y6 Y7 Y8 Y9 Y10 Cash And Equivalents 24,774.50 15,229.80 19,150.60 18,738.10 25,323.80 15,482.00 21,597.80 18,918.50 18,342.00...
-
JenCo reported the following information in its financial statements for three successive quarters during the 2021 fiscal year ($ in millions): Balance sheets: Three Months Ended 2021/05/01 (Q3)...
-
Feather Friends, Incorporated, distributes a high-quality wooden birdhouse that sells for $24 per unit. Variable expenses are $12 per unit, and fixed expenses total $200,000 per year. Its operating...

Study smarter with the SolutionInn App


