From models of the transition states for their reactions, predict which of the following two diastereomers of
Question:
From models of the transition states for their reactions, predict which of the following two diastereomers of 3-bromo-2-butanol should form an epoxide at the greater rate when treated with base, and explain your reasoning.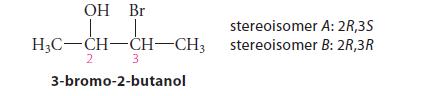
Transcribed Image Text:
OH Br H₂C-CH-CH-CH3 3 3-bromo-2-butanol 2 stereoisomer stereoisomer A: 2R,3S B: 2R,3R
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The key to solving this problem is to realize that in order for epoxide formation to occur the oxyge...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When the first compound shown here is treated with sodium methoxide, the only elimination product is the trans isomer. The second diastereomer (blue) gives only the cis product. Use your models and...
-
Two of the compounds given in Fig. Pl 1.78 form epoxides readily when treated with -OH, one forms an epoxid slowly, and one does not form an epoxide at all. Identify the compound(s) in each category...
-
Justin Bieber has emerged as a bona fide sports star, but in the most unlikely of sportstable tennis. At only 24 years old, Bieber has won 8 of the last 11 major table tennis tournaments over the...
-
Gems Co. uses the indirect method to prepare its statement of cash flows. The following comparative statement of financial position for 2021 and 2022 are presented: At December 31 2022 2021 Property,...
-
Information from comparative income statements and balance sheets for Microsoft and IBM is given below. (Amounts are in millions.) Use this information to answer the following questions: 1. Without...
-
Identify and briefly describe five common fact-finding methods.
-
Suppose \(\mathbf{x} \sim M N(n, \boldsymbol{\pi})\) follows a multinomial distribution of size \(n\) and probability \(\pi\). Derive the variance matrix of \(\mathbf{x}\).
-
Lee Werner is general manager of Stoneybrook Salons. During 2016, Werner worked for the company all year at a $ 14,000 monthly salary. He also earned a year-end bonus equal to 15% of his annual...
-
Database design is quite complex and important. Discuss the role played by users during the design process Describe the purpose of normalizing data Briefly describe the basic SQL DDL statements and...
-
The chlorohydrin trans-2-chlorocyclohexanol reacts rapidly in base to form an epoxide. The cis stereoisomer, however, is relatively unreactive and does not give an epoxide. Explain why the two...
-
Give the product expected when each of the following alkenes is treated with MMPP. (a) trans-3-hexene (b) =CH
-
Mason Advertising Agency was founded in January 2008. Presented below are adjusted and unadjusted trial balances as of December 31, 2012. Instructions(a) Journalize the annual adjusting entries that...
-
Research the current state of the companys market and how theyve done in a predicted post-pandemic boom. Come to class prepared to discuss events beyond the time period covered in this chapter.
-
Are you concerned by any of the data-use possibilities that you outlined in prior questions, discussed in this chapter, or that youve otherwise read about or encountered? If you are concerned, why?...
-
What is meant by LIBOR and LIBID. Which is higher?
-
Consider sharing economy markets like ride-sharing and home rental. Have you or friends used the leading firms in these industries? Have you used firms that aren't the leaders? Why or why not?
-
Research this segment and identify firms that should be considered part of the sharing economy but that arent mentioned. What do you think of the prospects for the firm or firms youve identified? Why...
-
Buford purchased a new automobile in March for $23,000. In April, he receives a $500 rebate check from the manufacturer. The rebate was paid to all customers who purchased one of the manufacturer's...
-
Would you use the adjacency matrix structure or the adjacency list structure in each of the following cases? Justify your choice. a. The graph has 10,000 vertices and 20,000 edges, and it is...
-
Suggest a mechanism by which ?-ionone is transformed into ?-ionone on treatment with acid. H3o -Ionone B-Ionone
-
Draw the most stable chair conformation ofdihydrocarvone CH3 -H Dihydrocarvone
-
Draw the most stable chair conformation of menthol, and label each substituent as axial orequatorial. Menthol (from peppermint oil) - CH
-
Zara is about to introduce sustainable clothing line for first time in Pakistan. Explain Why do you think it is important to plan? Provide latest in text citations and harvard references.
-
Assume that the holding cost of inventory at Macys is 35% per year. Assume further that Macys turns its inventory 3 times per year. How much does it cost Macys to hold a dress that it purchased for...
-
Assume you established a new pastry shop. 1- How would you segment your customers (What may be the effective segmentation criteria?) 2- What segment would you target? Why? 3- How would you position...

Study smarter with the SolutionInn App


