From the outcome of the transformation shown in Fig. P6.48, indicate whether the levorotatory enantiomer of the
Question:
From the outcome of the transformation shown in Fig. P6.48, indicate whether the levorotatory enantiomer of the product has the R or S configuration. Draw a structure of the product that shows its absolute configuration.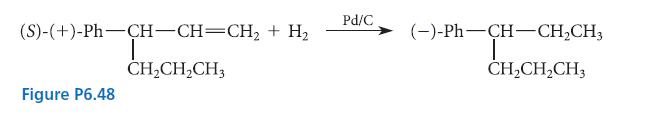
Transcribed Image Text:
(S)-(+)-Ph-CH-CH=CH₂ + H₂ I CH₂CH₂CH3 Figure P6.48 Pd/C (-)-Ph—CH-CH,CH, T CH₂CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
First draw the Senatiomer of the starting material using line ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Studies have shown that one stereoisomeric certain types of neurodegenerative disorders, system, as illustrated in structure B, and that form of compound A is an effective agent against Recognize...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The auditors report that follows was drafted by a staff accountant of Williams & Co., CPAs, at the completion of the audit of the financial statements of Lenz Corporation (nonpublic company) for the...
-
Assuming 10% minimum required reserve ratio, 5% excess reserve ratio, and people holding 10% of cash received at home (not deposited in the banks), the money multiplier is equal to Select one: a....
-
Suppose a defendant goes to a bar, gets blind drunk, and then goes out and robs a store. Is his intoxication a defense to the robbery?
-
The December 31, 20X6, condensed balance sheets of Pine Corporation and its 90 percent-owned subsidiary, Slim Corporation, are presented in the accompanying worksheet. Additional Information Pine's...
-
Which corporate characteristic is a disadvantage? a. Double taxation b. Mutual agency c. Limited liability d. None of the above
-
An analyst for an oil company has developed a formal linear regression model to predict the sales of 50 of their filling stations. The estimated model is Ŷ = b0 + b1 X1 where...
-
Calculate the incremental change to cash flow in the final year of this project. The project requires an initial investment of $382,000 for fixed assets and $83,000 for net working capital. The fixed...
-
(a) Explain why an optically inactive product is obtained when (2)-3-methyl-1-pentene undergoes catalytic hydrogenation. (b) What is the absolute configuration of (1)-3-methylhexane if catalytic...
-
(a) Give the stereochemical relationship (enantiomers, diastereomers, or the same molecule) between each pair of compounds in the set shown in Figure P6.46. Assume that internal rotation is rapid....
-
Using Table 14. 1 and Table 14. 2 from the text, calculate the current ratio, days cash on hand, days in accounts receivable, total asset turnover, interest coverage, long-term debt to net assets,...
-
The applied behavioural science of employee interactions and organizational processes within a business or work environment. Discuss?
-
Hannibal Steel Company's Transport Services Department provides trucks to haul ore from the company's mine to its two steel mills- the Northern Plant and the Southern Plant Budgeted costs for the...
-
Is Dawson subject to the provisions of the Hong Kong Prevention of Bribery Ordinance (POBO) in any business dealings outside of Hong Kong? Provide a brief explanation of your decision. If Dawson...
-
The Muffin House is an industrial bakery in Ogunquit, Maine. They produce baked goods in large batches. For each batch they record a range of information. A sample of this information is provided in...
-
A single-degree-of-freedom system experiences the periodic excitation in Fig. 1. The excitation is constant for time intervals of duration T/2 followed by time intervals also of duration T/2 during...
-
An oil company purchased an option on land in Alaska. Preliminary geologic studies assigned the following prior probabilities. P(high-quality oil) = .50 P(medium-quality oil) = .20 P(no oil) = .30 a....
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
An electrostatic potential map of (trifluoromethyl) benzene, C6H5CF3, is shown. Would you expect (trifluoromethyl) benzene to be more reactive or less reactive than toluene toward electrophilic...
-
Acetanilide is less reactive than aniline toward electrophilic substitution.Explain. c%3D0 Acetanilide CH
-
Draw resonance structures for the intermediates from reaction of an electrophile at the ortho, Meta and Para positions of nitrobenzene. Which intermediates are most stable?
-
What is the difference between brand marketing and direct marketing? When do you use brand marketing? When do you use direct marketing? Which can be measured and how? Name a company that makes...
-
Research and Create mini brand audit for Tim Hortons. Please expound/elaborate answers Mini Audit Categories Brand Name/Logo(s) Tagline/Slogan Top 3 competitors Canadian Market POP against #1...
-
Identify the 4P's of the Amazon Describe how the brand uses the marketing mix elements as a guide. Analyze the impact of the marketing mix on overall brand perception. Describe the role...

Study smarter with the SolutionInn App


