From what alkene and by which methods could you prepare each of the following alcohols essentially free
Question:
From what alkene and by which methods could you prepare each of the following alcohols essentially free of constitutional isomers?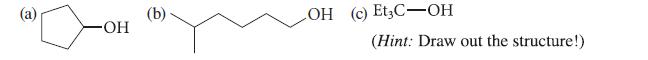
Transcribed Image Text:
(a) OH (b) LOH (c) Et3C-OH (Hint: Draw out the structure!)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a Because the OH is placed symmetrically in the alcohol either ...View the full answer

Answered By

Felix Onchweri
I have enough knowledge to handle different assignments and projects in the computing world. Besides, I can handle essays in different fields such as business and history. I can also handle both short and long research issues as per the requirements of the client. I believe in early delivery of orders so that the client has enough time to go through the work before submitting it. Am indeed the best option that any client that can think about.
4.50+
5+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following alcohols can be synthesizecl relatively free of constitutional isomers and diastereomers by oxymercuration-reduction? Explain. H,C CH,CH2CH CH2CH CH H,C H,C
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Suppose there are three buyers of candy in a market: Tex, Dex, and Rex. The market demand and the individual demands of Tex, Dex, and Rex are shown on the next page. a. Fill in the table for the...
-
Comfy Fit Company manufactures two types of university sweatshirts, the Swoop and the Rufus, with unit contribution margin s of $5 and $15, respectively. Regardless of type, each sweatshirt must be...
-
Why is defining the scope of a business alliance important?
-
Although the customer loyalty project at Petrie Electronics had gone slowly at first, the past few weeks had been fast-paced and busy, Jim Watanabe, the project manager, thought to himself. He had...
-
The Bayside Art Gallery is considering installing a video camera security system to reduce its insurance premiums. A diagram of the eight display rooms that Bayside uses for exhibitions is shown in...
-
IQ scores (as measured by the Stanford-Binet intelligence test) are normally distributed with a mean of 100 and a standard deviation of 16. What percentage of the population has an IQ score between...
-
Indicate whether each of the following reactions is homolytic or heterolytic, and tell how you know. Write the appropriate fishhook or curved-arrow notation for each. (a) :N=C:+ CH,CH, CHCH3 I :Br:...
-
Ozonolysis of 2-pentene gives a mixture of the following three ozonides. Using the mechanism in Eqs. 5.36a and 5.36b, explain the origin of all three ozonides. HC-HC A CH-CH3 HC-HC B CH-CHCH3...
-
How can a firm hedge long term currency positions? Elaborate on each method.
-
Problem 1 Below you find mortgage rate information: Fixed Rate Mortgages Term Posted Rate (APR) 5 year 5.29% 6 year 6.16% 6.35% 6.75% 7 year 10 year Assume that the bank calculates monthly payments...
-
Linda bough 500 CIBC shares worth of $10000 on April 1st in her son's name. her son brad is 16 years old, CIBC pays $2000 (eligible) dividend income every year. Who should report dividend income?...
-
Which type of relational maintenance strategy is invoked when a romantic partner buys an engagement ring for the other partner? assurances openness understanding positivity.
-
Tom purchases a property and finances it with a Graduated Payment Mortgage (GPM) loan for 200,000 at j 2 =7% (2 is compounding frequency) His monthly payment for the first year will be 1000; the...
-
A football is thrown upward at a(n) 36 angle to the horizontal. The acceleration of gravity is 9.8 m/s. To throw a(n) 66.2 m pass, what must be the initial speed of the ball? Answer in units of m/s.
-
The comparative balance sheets for 2011 and 2010 are given below for Surmise Company. Net income for 2011 was $50 million. Required: Prepare the statement of cash flows of Surmise Company for the...
-
You continue to work in the corporate office for a nationwide convenience store franchise that operates nearly 10,000 stores. The per- store daily customer count (i.e., the mean number of customers...
-
Show the products of these acid-base reactions and predict whether the equilibria favor the reactants or the products: a) CHCCHCCHCH + OCHCH b) CHCHNO + CHO: CH3 (c) CH3COCH, + CHCH- 10 1:Z: LL CH,...
-
Which compound is behaving as the Lewis acid and which as the Lewis base in this reaction? AICI3 T CHCHCHCH3 + AICI CHCHCHCH3
-
Explain which of these compounds is the weaker base? :Z 0 HIN:
-
If a projectile with a mass of 9.30 g is traveling at a speed of 1.26 km/s, determine the following. (a) the kinetic energy (in kJ) of the projectile in kilojoules kJ (b) the kinetic energy (in kJ)...
-
13. You are part of a student engineering team competing in a flywheel-car race. When the race starts, each team can begin winding a flywheel that will then propel their car towards a finish line....
-
3. (25 points) A wave in the ocean has a wave height of 3.80 meters and a wave period of 14.5 seconds. The wave is propagating through the ocean, and at a depth of 300 meters, the wave angle of...

Study smarter with the SolutionInn App


