Give all of the product(s) expected, including pertinent stereochemistry, when each of the following compounds reacts with
Question:
Give all of the product(s) expected, including pertinent stereochemistry, when each of the following compounds reacts with sodium ethoxide in ethanol. (D = deuterium = 2H, an isotope of hydrogen.)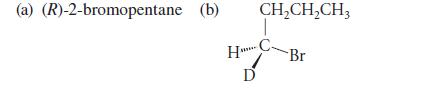
Transcribed Image Text:
(a) (R)-2-bromopentane (b) CH₂CH₂CH3 HBr C D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a CHCHCH3 CH...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the mechanistic symbols (SN1, SN2, E1, E2) that are most consistent with each of the following statements: (a) Methyl halides react with sodium ethoxide in ethanol only by this mechanism. (b)...
-
Give the structure of the expected product from the reaction of isopropyl benzene with (a) Hydrogen (3 mol), Pt (b) Sodium and ethanol in liquid ammonia (c) Sodium dichromate, water, sulfuric acid,...
-
Give the structure of the expected product from the reaction of isopropylbenzene with (a) Hydrogen (3 mol), Pt (b) Sodium and ethanol in liquid ammonia (c) Sodium dichromate, water, sulfuric acid,...
-
A long, slender bar in the shape of a right circular cone with length L and base diameter d hangs vertically under the action of its own weight (see figure). The weight of the cone is W and the...
-
The Town of McHenry has $6,000,000 in general obligation bonds outstanding and maintains a single debt service fund for all debt service transactions. On July 1, 2012, a current refunding took place...
-
The capital investment committee of Touch of Eden Landscaping Company is considering two capital investments. The estimated income from operations and net cash flows from each investment are as...
-
Consider the following cash flow profile, and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Designing a Balanced Scorecard for a pharmaceutical company Chadwick, Inc.: The Balanced Scorecard (Abridged)14 Company Background Chadwick, Inc., was a diversified producer of personal consumer...
-
According to the Civil Code of the Philippines, Article 1 7 6 7 defines partnership as: By the contract of partnership two or more persons bind themselves to contribute money, property, or industry...
-
In the Williamson ether synthesis, an alkoxide reacts with an alkyl halide to give an ether. You are in charge of a research group for a large company, Ethers Unlimited, and you have been assigned...
-
In each of the following series, order the atoms, compounds, or ions in order of increasing polarizability, and explain your choices. (a) Se, O, S (b) Chloroform, fluoroform, iodoform (c) I , Br ,...
-
Sunshine Solar, Inc., had Sales Revenue of $470,000, Sales Returns and Allowances of $19,000, and Sales Discounts of $6,000 in 2016. Cost of goods sold was $193,000, and net income was $96,000 for...
-
Distinguish between a job description and a job specification.
-
What is meant by the terms personality and proactive personality?
-
Discuss the issue of fairness and an ethical approach in employee selection.
-
Comment on graphology as a selection method.
-
At the end of the financial year, prepare the general journal to close out the ledger accounts to the trading account in the general ledger. Post the journal to the T accounts shown in figure 9.9....
-
Rockyford Company must replace some machinery that has zero book value and a current market value of $1,800. One possibility is to invest in new machinery costing $40,000. This new machinery would...
-
What kind of rays are X-rays?
-
Compound A, C 11 H 16 0, was found to be an optically active alcohol. Despite its apparent unsaturation, no hydrogen was absorbed on catalytic reduction over a palladium catalyst. On treatment of A...
-
One of the steps in fat metabolism is the hydration of crotonate to yield 3-hydroxybutyratc. The reaction occurs by addition of OH to the Si face at C3, followed by Protonation at C2, also from the...
-
The dehydration of citrate to yield cis-aconitate, a step in the citric acid cycle, involves the pro-R ?arm? of citrate rather than the pro-S arm. W1ich of the following two products is formed? ...
-
When a business becomes insolvent, it Must liquidate enough assets to pay all creditors the full amount they were owed before it can be reorganized. Can be liquidated or reorganized. Must be...
-
As part of the reorganization plan, the loan is exchanged for a new loan in the amount of $820,000 plus 35% of the new stock issued. The note is exchanged for a new note in the amount of $1,600,000...
-
Record the value of angles and fill up DH table accordingly) Axis (i) 01 341 d 2 3 4 5 6 a=the distance from Z., to Z, measured along X. the angle from Z., to Z, measured about X = d, the distance...

Study smarter with the SolutionInn App


