Give the product(s) expected when d-mannose (or other compound indicated) reacts with each of the following reagents.
Question:
Give the product(s) expected when d-mannose (or other compound indicated) reacts with each of the following reagents. (Assume that cyclic mannose derivatives are pyranoses.)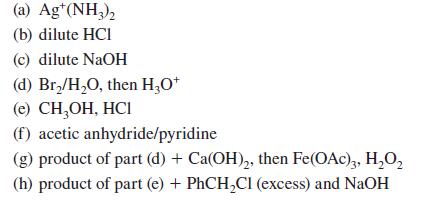
Transcribed Image Text:
(a) Ag¹(NH3)₂ (b) dilute HCI (c) dilute NaOH (d) Br₂/H₂O, then H3O+ (e) CH₂OH, HCI (f) acetic anhydride/pyridine (g) product of part (d) + Ca(OH)₂, then Fe(OAc)3, H₂O₂ (h) product of part (e) + PhCH₂Cl (excess) and NaOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Note that carboxylic acids derived from carbohydrates usually exist as l...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the principal organic product(s) expected when 2- methylthiophene or other compound indicated reacts with each of the following reagents. (a) HNO3 (b) Dilute aqueous NaOH (c) Product of part (a)...
-
Give the principal organic product(s) expected when p-chloroaniline or other compound indicated reacts with each of the following reagents. (a) Dilute HBr (b) CH 3 CH 2 MgBr in ether (c) NaNO 2 ,...
-
Give the principal organic product(s) expected when 2- methylpyridine or other compound indicated reacts with each of the following reagents. (a) Diliute aqueous NaOH (b) HNO3, H2SO4, heat; then -OH...
-
Write a nonrecursive function that takes the first Node in a linked list as an argument and reverses the list, returning the first Node in the result.
-
Give the symbols for two other isotopes of (a) 14 N, (b) 56 Fe, and (c) 118 Sn
-
What are some of the major reasons for the rapid expansion in international mergers and joint ventures of firms?
-
Prove that if \[d r_{t}^{i}=\left(\delta_{i}-k r_{t}^{i} ight) d t+\sigma \sqrt{r_{t}^{i}} d W_{t}^{i}, i=1,2\] where \(W^{i}\) are independent BMs, then the sum \(r^{1}+r^{2}\) is a CIR process....
-
Jennifer Capriati Corp. has a deferred tax asset account with a balance of $150,000 at the end of 2007 due to a single cumulative temporary difference of $375,000. At the end of 2008 this same...
-
A lab project involves selecting between two experimental methods: Method A, which has a 70% success rate, and Method B, with a 90% success rate. If Method A costs $5,000 and Method B costs $8,000,...
-
Give the products expected when D-ribose (or other compound indicated) reacts with each of the following reagents. (a) dilute HNO3 (b) -CN, HO (c) product of part (b) + H/Pd/BaSO4 + HO+/HO (d) CHOH,...
-
Consider the structure of cellobiose, a disaccharide obtained from the hydrolysis of the polysaccharide cellulose. Into what monosaccharide(s) is cellobiose hydrolyzed by aqueous HCl? HO CHOH -...
-
In its most recent annual report, Laurentian Beverages reported current assets of $54,000 and a current ratio of 1.80. Assume that the following transactions were completed: a. Purchased merchandise...
-
What is your basic attitude toward the realities you face? Is your reality a gift or an obstacle, a friend or an enemy?
-
Are the leadership and management development programs provided to our managers well equipped to help develop the skills and competences needed to adequately employ an integrative responsible...
-
A different perspective may be observed from the #Metoo movement, as it can be examined as a manifestation of leading, with the potential to challenge our understanding of leadership. Could #Metoo be...
-
How can you incorporate mindfulness practice and training into your personal and organizational business routines?
-
Think of an organization of your choice in parastatal stage of transformation. What are the major challenges that managers have to deal with? What are real or potential benefits of Hybrid/In-Between...
-
The Conference Board published information on why companies expect to increase the number of part-time jobs and reduce full-time positions. Eighty-one percent of the companies said the reason was to...
-
In Exercises 516, find the focus and directrix of the parabola with the given equation. Then graph the parabola. y 2 = 4x
-
When DNA is treated with 0.5 M NaOH at 25C, no reaction takes place, but when RNA is subjected to the same conditions, it is rapidly cleaved into mononucleotide 2- and 3-phosphates. Explain.
-
Indicate whether you would expect imidazole to have high or low water solubility, and why.
-
What product is expected when 4-(dimethylamino)pyridine reacts with CH3I?
-
What kinds of things do you need to do while planning to ensure that you can measure using EV (earned value) later during execution? Are there processes that need to be added during planning? Are...
-
what ways can ethical leadership models, such as servant leadership or authentic leadership, cultivate a culture of integrity by inspiring trust, promoting transparency, and fostering accountability...
-
Determine how a responsible firm should make these trade-offs and explain your position. Also, create a ranked list that identifies what steps can strategic leaders take to guide organizations on...

Study smarter with the SolutionInn App


