Give the products expected when D-ribose (or other compound indicated) reacts with each of the following reagents.
Question:
Give the products expected when D-ribose (or other compound indicated) reacts with each of the following reagents.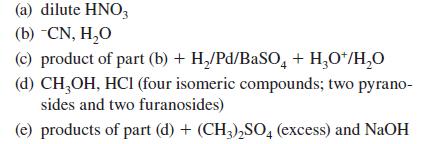
Transcribed Image Text:
(a) dilute HNO3 (b) -CN, H₂O (c) product of part (b) + H₂/Pd/BaSO4 + H₂O+/H₂O (d) CH₂OH, HCI (four isomeric compounds; two pyrano- sides and two furanosides) (e) products of part (d) + (CH3)₂SO4 (excess) and NaOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Note that carboxylic acids derived from carbohydrates usually exist as lactones and aldos...View the full answer

Answered By

Isaiah Mutinda
As a graduate with Bs in Maths and Computer Science and having worked as a freelance full stack software developer for 3 years running I believe I have what it takes to conformable tutor and mentor a student to a professional developer also.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the principal organic product(s) expected when 2- methylthiophene or other compound indicated reacts with each of the following reagents. (a) HNO3 (b) Dilute aqueous NaOH (c) Product of part (a)...
-
Give the principal organic product(s) expected when 2- methylpyridine or other compound indicated reacts with each of the following reagents. (a) Diliute aqueous NaOH (b) HNO3, H2SO4, heat; then -OH...
-
Give the principal organic product(s) expected when ethyl benzoate or the other compound indicated reacts with each of the following reagents. (a) HO, heat, acid catalyst (b) NaOH, HO (c) aqueous...
-
In the game of chess, if a board position is repeated three times with the same side to move, the side to move can declare a draw. Describe how you could test this condition using a computer program.
-
Calculate the binding energy and the binding energy per nucleon from the masses given in Table 40-1 for (a) 12 C, (b) 56 Fe, (c) 238 U. Table 40-1 Atomic Masses of the Neutron and Selected Isotopes...
-
Match the following and select the correct option. Height of Binary search tree(in worst o([log ]-1) 1 a (2n+1) case) Height of Ternary tree b O(n) Height of B-tree(when c minimum degree, -2) (log,...
-
The normalized Brownian excursion \(|B|^{\left[g_{1}, d_{1} ight]}\) is a \(\mathrm{BES}^{3}\) bridge from \(x=0\) to \(y=0\) with \(t=1\). \[\begin{equation*} X_{t}^{[a, b]}=\frac{1}{\sqrt{b-a}}...
-
Honeybutter, Inc., manufactures a product that goes through two departments prior to completionthe Mixing Department followed by the Packaging Department. The following information is available about...
-
A lab project involves three critical tasks: Experiment Setup, Data Collection, and Analysis. Task 1 takes 8 days with a variance of 1, Task 2 takes 12 days with a variance of 4, and Task 3 takes 10...
-
What product(s) would be obtained when cellulose is treated first exhaustively with dimethyl sulfate/NaOH, then with 1 M aqueous HCl?
-
Give the product(s) expected when d-mannose (or other compound indicated) reacts with each of the following reagents. (Assume that cyclic mannose derivatives are pyranoses.) (a) Ag(NH3) (b) dilute...
-
Stuart Scroggins died in June of the current year. Among the provisions in his will is one that benefits your client, Samantha Scroggins Valdez. This provision reads as follows: I leave $1.5 million...
-
Metaphors about barriers to women leadership abound. In your view, which metaphor is the most representative of such barriers? And why?
-
Think of VUCA. Give an example of an organization that has successfully confronted transformational forces of VUCA. Can you recognize the power of mission or vision in this transformation?
-
Think of ambidexterity. What kind of structural configurations, organizational cultures, and leadership styles are in best position of boosting ambidexterity in VUCA environments?
-
Are you mindful of the necessity for crisis preparedness in your role in the organizations you work in?
-
How might decision-makers creatively change their mental models of higher education to make colleges and universities more assessable, affordable, and achievable to historically marginalized...
-
According to the U.S. Bureau of the Census, about 75% of commuters in the United States drive to work alone. Suppose 150 U.S. commuters are randomly sampled. a. What is the probability that fewer...
-
In Exercises 105108, evaluate each expression without using a calculator. log(ln e)
-
Although the synthesis of heterocyclic rings was not discussed in the text, many such syntheses employ reactions that are similar or identical to reactions in other parts of the text. Give...
-
One theory of genetic mutation postulates that some mutations arise as the result of mispairing of bases in DNA caused by the existence of relatively rare isomeric forms of the bases. Show the...
-
The stability of a DNA double helix can be measured by its melting temperature, Tm, defined as the temperature at which the helix is 50% dissociated into individual chains. Which of the following...
-
What are your three most important personal and professional priorities regarding the completion of your dissertation research and life beyond graduation? Why are these important to you and the...
-
Please make sure to fill every box. Once completed, you will have defined two long-term goals with related medium- and short-term goals. Defining Goals Worksheet GOAL PRIORITIES MY PRECISE GOALS...
-
What circumstances/factors can cause an individual disputes to escalate to the collective level? Is a worker an employee? What role does the state play in employee relations and how is this role...

Study smarter with the SolutionInn App


