Give the structure of the diene and dienophile that would react in a DielsAlder reaction to give
Question:
Give the structure of the diene and dienophile that would react in a Diels–Alder reaction to give the following product: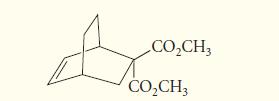
Transcribed Image Text:
CO₂CH3 CO₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
In the product of a DielsAlder reaction the two carbons of the double bond and the two adjacent carb...View the full answer

Answered By

Caroline Kinuthia
Taking care of the smaller details in life has a larger impact in our general well being, and that is what i believe in. My name is Carol. Writing is my passion. To me, doing a task is one thing, and delivering results from the task is another thing. I am a perfectionist who always take things seriously and deliver to the best of my knowledge.
4.90+
1933+ Reviews
4269+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the diene and dienophile that would react in a DielsAlder reaction to give each of the following products. (a) CN (b) COCH3
-
Give the diere and dienophile that would react in a Diels-Alderreaction to give each of the following products. CN
-
Waterways Corporation uses very stringent standard costs in evaluating its manufacturing efficiency. These standards are not "ideal" at this point, but the management is working toward that as a...
-
Select only 2. Two questions which can be used to address ethical issues impacting organizations and Human Resource Management include: What would my Supervisor say? Does this behavior or result meet...
-
What does the term deferral mean?
-
Janetta Corp. has an EBIT rate of $975,000 per year that is expected to continue in perpetuity. The unlevered cost of equity for the company is 14 percent, and the corporate tax rate is 35 percent....
-
On December 14, 2011, appellant Aaron Olson contracted to receive telephone service from respondent CenturyLink and also applied for reduced-rate service that CenturyLink provides through Minnesotas...
-
Venice InLine, Inc., was founded by Russ Perez to produce a specialized in-line skate he had designed for doing aerial tricks. Up to this point, Russ has financed the company with his own sayings and...
-
What ways can I make my Relational Data Model conversion meet the following requirements listed below? Am I on the right track? What corrections do I need to address? ERD: My Conversion of ERD onto a...
-
Which of the following carbocations is more stable? + CH0-CH=CH-CH or A :CH, 1 HC=C-CH B +
-
An optically active alkyne A (C 10 H 14 ) can be catalytically hydrogenated to butylcyclohexane. Treatment of A with EtMgBr liberates no gas. Catalytic hydrogenation of A over Pd/C in the presence of...
-
A laboratory container is shaped like the frustum of a cone: Write methods to compute the volume and surface area, using these equations: R. R1
-
List the most important tools for tracking online activities. How are these tools used?
-
In general, how is the rate on a floating-rate bond determined?
-
Do you think any social media companies manipulate consumers? Why or why not? If so, which ones?
-
What type of information is provided through the analysis of digital tracking data?
-
Which is riskier to an investor, other things held constanta callable bond or a putable bond? Explain.
-
Lucinda, a welder for Big Auto, Inc., dies in an automobile accident on March 14 of this year. Big Auto has a company policy of paying $5,000 to the spouse of any employee who dies. In addition to...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
A knowledge of molar absorptivities is particularly important in biochemistry, where UV spectroscopy can provide an extremely sensitive method of analysis. For example, imagine that you wanted to...
-
Which of the following compounds would you expect to show ultraviolet absorptions in the 200 to 400nmrange? (a) (b) (c) CN CH3 (f) (d) (e) " N. Indole Aspirin
-
Show the structures of all possible adducts of the following diene with 1 equivalent ofHC1:
-
Rod Bullhorn is a resident of Ohio. Mr. Bullhorn purchased an Ohiomunicipal bond. What is the tax treatment of the interest that Rod earns on his Ohio bond?
-
The cost, C, of air conditioning your home, in dollars per day, when the temperature outside is T degrees Fahrenheit is given by y = C(T). When T = 92F, C(92) = 1.83 and C'(T) = 1.64, for 90F
-
The Sloan Corporation is trying to choose between the following two mutually exclusive design projects: Cash Flow Cash Flow Year (1) (II) 0723 -$62,000-$18,200 1 33,000 9,800 33,000 9,800 33,000...

Study smarter with the SolutionInn App


