Given the boiling point of the first compound in each set, estimate the boiling point of the
Question:
Given the boiling point of the first compound in each set, estimate the boiling point of the second.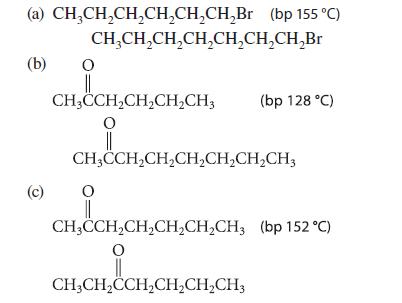
Transcribed Image Text:
(a) CH₂CH₂CH₂CH₂CH₂CH₂Br (bp 155 °C) (b) (c) CH₂CH₂CH₂CH₂CH₂CH₂CH₂Br O CH₂CCH₂CH₂CH₂CH3 2CH₂CH O || CH3CCH₂CH₂CH₂CH₂CH₂CH3 O (bp 128 °C) CH3CCH₂CH₂CH₂CH₂CH3 (bp 152 °C) O CH3CH₂CCH₂CH₂CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Use the rule of thumb that an additional carbon ad...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
TIME 1 2 8 AM The following data were collected on the amount of deodorant in cans, in grams, filled in the filling line of a packaging company. Prepare X- and R-charts for the data, and comment on...
-
Estimate the boiling point of a 0.050 m aqueous MgCl2 solution. Assume a value of i based on the formula.
-
Glorious Electrical Appliances (GEP) Co. is a company that sells electrical tools. GEP uses perpetual inventory system in recording its inventory. The financial position of GEP as at 31 December 2016...
-
The Federal Reserve may raise its benchmark interest rate later this month. How is this achieved? Why would they do this? Explain the consequences fully. (Include graphs with your answer)
-
Multiple Choice 1. Colorado passes a hotel tax of 8 percent for Colorado residents and 15 percent for out-of-state visitors. The new law A. Is valid, based on the Supremacy Clause B. Is void, based...
-
Stratford Corporation is a diversified company whose products are marketed both domestically and internationally. Its major product lines are pharmaceutical products, sports equipment, and household...
-
What is the difference between a predator and a situational (accidental) fraudster?
-
Teri Hall has recently opened Sheer Elegance, Inc., a store specializing in fashionable stockings. Ms. Hall has just completed a course in managerial accounting, and she believes that she can apply...
-
Prove that if M is a dense linear subspace of a separable Hilbert space H, then H has an orthonormal basis consisting of elements in M. Does the same result hold for arbitrary dense subsets of H?
-
Draw the structures and give the names of all isomers of octane with (a) Five carbons (b) Six carbons in their principal chains.
-
Give a general balanced reaction for (a) The complete combustion of an alkane (formula C n H 2n+2 ). (b) The complete combustion of a cycloalkane containing one ring (formula C n H 2n ).
-
Explain why resources are allocated efficiently among firms and why output is distributed efficiently among households in perfectly competitive markets.
-
For the following REDOX reaction, state the oxidation number for each element in the reaction. State which element is being oxidized and which element is being reduced, as well as how you know. State...
-
Common stock with a total par value of $80,000 is issued at a total of $110,000. The $30,000 excess above the par value should be recorded as a CREDIT to which account?
-
Clausewitz used this term to describe those assets key to an enemy's ability to survive and carry out warfare. A successful belligerent can attack and seize or destroy these assets. Examples of such...
-
What are the ecological implications of trophic interactions and food chain dynamics for ecosystem services provisioning, including the regulation of pest populations, pollination services, nutrient...
-
Explain the difference between statutory law, common law, and business law. ?
-
What are economic resources? What categories do economists use to classify them? Why are resources also called factors of production? Why are they called inputs?
-
How can a promoter avoid personal liability for pre-incorporation contracts?
-
Predict the products of the reaction of (i) phenyl acetaldehyde and (ii) acetophenone with the following reagents: (a) NaBH4 then H3O+ (b) Tollens reagent (c) NH2OH, HC1 catalyst (d) CH3MgBr, then...
-
How would you prepare the following substances from 2-cyclohexcnone? More than one step may be required. .CH (a) (b) (d) (c) CSH5 (Two ways)
-
Show how the Wittig reaction might be used to prepare the following alkenes. Identify the alkyl halide and the carbonyl components that would he used. (b) (a)
-
The city of Dilworth spends $ 1 0 0 , 0 0 0 a year on janitorial services for city buildings. Rather than set up a separate funding source for janitorial services, the city would prefer to pass along...
-
Question: Question 8 ?of 10\table[[Number,April,May,June,Completed],[10,$4,860,$4,220,,May],[11,3,840,3,680,$1,880,June],[12,1,100,,,April],[13,,4,390,4,230,June],[14,,5,560,3,370,Not complete]]Job...
-
Using the following facts, calculate what total revenue on Part VIII, Statement of Revenue of IRS Form 990 would be for this example social service organization. You just need to calculate the total...

Study smarter with the SolutionInn App


