In each case, give the structure of a starting material that would give the product shown by
Question:
In each case, give the structure of a starting material that would give the product shown by MnO2 oxidation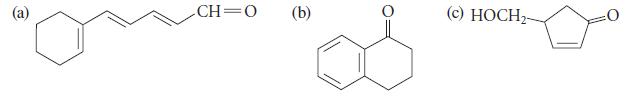
Transcribed Image Text:
@ CH=O (b) O (c) HOCH₂-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a CH...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Refer to Study Problem 28.2 and, assuming an antarafacial migration, give the structure of a starting material that would give a stereoisomer of the product with the R configuration at the...
-
Give the structure of the product expected when each of the following alcohols is subjected to MnO2 oxidation. (a) (b) CH2OH CHOH OH
-
Give the structure of the product that would result from a Baeyer-Villiger oxidation of cyclopentanone.
-
Can you draw the upper shear zone margin on Figure 16.9? Is it easily definable?
-
Calculating and interpreting cash flow from operations The following items appear in the financial statements of Bamberger Enterprises a firm offering IT services for Sarbanes-Oxley compliance, for a...
-
Martin Service Company began the year 2010 with cash of $55,900. In addition to earning a net income of $38,000 and making cash withdrawals of $19,500, Martin Service borrowed $78,000 from the bank...
-
If a \({ }_{92}^{238} \mathrm{U}\) nucleus is struck by a neutron, it may absorb the neutron. The resulting nucleus then rapidly undergoes betaminus decay. The daughter nucleus of that decay is A....
-
The West Division of Nieto Company reported the following data for the current year. Sales ............... $3,000,000 Variable costs .......... 1,950,000 Controllable fixed costs ....... 600,000...
-
Questions to be used as subheadings of your project. This project requires you to develop a play experience that you could implement into the service that promotes learning and is appropriate for...
-
What is the major CyP450-promoted hydroxylation product of each of the following compounds? Assume that benzylic hydroxylation occurs, and neglect stereochemistry. (a) toluene (b) N NMe imipramine...
-
Predict the major product that is obtained when each of the following alkyl halides is treated with potassium tert- butoxide. Explain your reasoning. (a) -Br (b) OCH3 T CH-CHI
-
Eight years ago, Burt Brownlee purchased a U.S. government bond that pays 3.80 percent interest. The face value of the bond was $1,000. a. What is the dollar amount of annual interest that Burt...
-
The maximum amount ofc apital expenditures eligible for immediate expensing is: a. $3,500,000 b. $25,000 c. $1000,000 d. $2,500,000
-
In the current year, James Jar Co., a calendar year taxpayer, incurred the following research and experimentation expenditures (R&E): Salaries$ 150,000; and Materials$ 100,000. a. If James Jar Co....
-
Jim, a retail merchant, reported the following on his 2018 return: What amount must be omitted from income for the six-year statute of limitations to apply? Sales Cost of goods sold Gross profit...
-
What qualifications must be met before a taxpayer is allowed a home office deduction?
-
Monique has just returned from her first business trip to Japan. While there, she was impressed with the intense use of work teams. Now back in Quebec, she would like to totally reorganize the...
-
A large multinational banking company wants to determine whether there is a significant difference in the average dollar amounts purchased by users of different types of credit cards. Among the...
-
Refer to the Conservation Ecology (Dec. 2003) study of the causes of forest fragmentation, presented in Exercise 2.166 (p. 97). Recall that the researchers used advanced high-resolution satellite...
-
Analyze the splitting pattern for a set of equivalent b protons in the presence of three equivalent adjacent a protons. Include an analysis of the relative intensity of each line of the splitting...
-
Explain why the following two structures are ruled out by the data in Study Problem 13.3. Problem 13.3 An NMR spectrum of a compound X contains four absorptions at δ 1.3, δ...
-
What is the reduction in shielding of Protons b relative to the protons of TMS in magnetic field units of gauss? The applied field in a 300 MHz NMR spectrometer is 70,500 gauss.
-
What questions come from The Coahuila y Texas Immigration Law of 1825 raise?
-
Which assumption permits businesses to record property and equipment as assets at their cost without having to be concerned about what they are worth in case of liquidation in the near future?
-
WHAT IS THE ROLE OF GLOBALIZATION IN INDIA'S DEVELOPMENT ?

Study smarter with the SolutionInn App


