Predict the major product that is obtained when each of the following alkyl halides is treated with
Question:
Predict the major product that is obtained when each of the following alkyl halides is treated with potassium tert- butoxide. Explain your reasoning.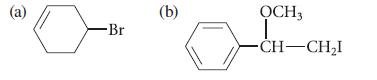
Transcribed Image Text:
(a) -Br (b) OCH3 T CH-CH₂I
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a b Although this alkyl halide has two chemically nonequivalent types of 3hydrogens an all...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product that is obtained when the following alkyl halides is treated with potassium terf-butoxide. Explain your reasoning. OCH CH CHI
-
Predict the major product obtained when each of the following compounds is treated with bromine (Br 2 ) together with sodium hydroxide (NaOH) followed by aqueous acid (H 3 O + ). (a) (b) (c)
-
Predict the major product obtained when each of the following compounds is treated with Birch conditions. a. b. c. d. e. f. OH
-
How can a layer fold and boudinage (extend) at the same time?
-
Preparing a statement of cash flows from changes in balance sheet accounts The comparative balance sheets of Southwest Airlines show the following information for a recent year (amounts in...
-
Below is information from the statement of owners equity of Mrs. Kittys Cookies for a recent year. Withdrawals ............. 0 Net income .............. ? Owners Equity, January 31, 2010...
-
Because the decay products in the above fission reaction are neutron rich, they will likely decay by what process? A. Alpha decay B. Beta decay C. Gamma decay The uranium isotope \({ }^{235}...
-
On January 1, 2017, Task Co. signs an agreement to lease office equipment from Coleman Inc. for three years with payments of $193,357 beginning December 31, 2017. The equipment's fair value is...
-
The final section of the Capstone Paper must review the challenge and recommend a strategy for addressing the challenge based on research. Along with the strategy, define the potential financial or...
-
In each case, give the structure of a starting material that would give the product shown by MnO 2 oxidation @ CH=O (b) O (c) HOCH-
-
What product(s) are formed when a Grignard reagent prepared from each of the following alkyl halides is treated with D 2 O? CHBr 1-(bromomethyl)cyclohexene (b) CH3 Br 6-bromo-1-methylcyclohexene
-
Using the information in Applied Problem 4, what decision would Remox make using each of the following rules if it had no idea of the probability of a tariff? a. Maximax b. Maximin c. Minimax regret...
-
LB Corporation purchased a business car in June 2019 for $62,000. The car weighs 4,000 pounds and will be used 100 percent of the time in the business. Compute the largest depreciation deduction...
-
What is active participation?
-
In March, Gary Parker started a new career as a real estate agent. He established Gary Parker Company, LLC on March 11 and began seeking clients. He listed his first property on March 21, and closed...
-
How can nonrental undertakings be combined?
-
Assume for the moment that you have a great idea for a potential Internet-based start-up business. In discussing the idea with a friend, she advises you to be very careful to tie your business idea...
-
Southwest Airlines is able to keep fares low, in part because of relatively low maintenance costs on its airplanes. One of the main reasons for the low maintenance costs is that Southwest flies only...
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
Predict the complete NMR spectrum of 1, 2-dichloropropane under each of the following assumptions. Note that protons and Hb and Hc are diastereotopic. Jab = Jac) Ha Hb HC-C-C-Cl ClHc...
-
Identify the following two isomeric alkyl halides (C5H11Br) from their 300-MHz NMR spectra, which are as follows: Compound A: 0.91 (6/7, d, J = 6 Hz); 1.7-1.8 (3H, complex); 3.42 (2H, t, J = 6 Hz)...
-
Explain how the NMR spectra of 1, 2, 2-trimethyl-1-propanol would change following a D20 shake.
-
On the first day of the year, Erin deposited $2500 into a savings account that earns 4% annual interest, compounded monthly. If she does not have any plans to deposit or withdraw any money, how much...
-
Whether there are other humanitarian groups that should be considered for protection in our immigration laws. If so, who?
-
Accountants use the worksheet to organize and complete adjustments for all but one of the following events: Depreciation on equipment. Revenue eamed and recorded in the current period. Supplies on...

Study smarter with the SolutionInn App


