Name the following compounds and tell whether each is a bridged or a fused bicyclic compound. (a)
Question:
Name the following compounds and tell whether each is a bridged or a fused bicyclic compound.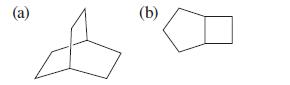
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Bicyclo222octane ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Without drawing their structures, tell which of the following compounds is a fused bicyclic compound and which is a bridged bicyclic compound, and how you know. bicyclo[2.1.1]hexane (A)...
-
Give the common name for each of the following compounds, and tell whether each is a primary, secondary, or tertiary alkyl halide. (a) (CH3)CHCH-F -Br (d) (b) CH3CHCHCHCHCH2-I CH3 T HC-C-CHCl I CH3
-
Name the following compounds, and indicate whether each is a reducing sugar or a nonreducing sugar: a. b. c. d. CH,OH HO OCH2CH2CH3 OH HO CH2OH OCH3 - HOCH OCH2CH3 CH OH OH OH
-
Simplify the expression 2x + (x + 1) into a single x + 1 fraction. The numerator of your answer is: The denominator of your answer is:
-
1. What is money laundering? 2. How did the money laundering occur here? 3. If Laboyce did not commit the fraud, what crime did he commit?
-
Erin, a single taxpayer, has a taxable income of $103,000 in the current year before considering the following capital gains and losses: Short-term capital gain $ 3,000 Long-term capital gain 22,000...
-
The Foreign Corrupt Practices act can levy fines of more than five hundred million against companies our size that do not eliminate corruption in their operations abroad. Can you understand why the...
-
On June 12, 2002, Delta Corporation was awarded a $160,000 contract for testing a product. The contract consisted of $143,000 for labor and materials, and the remaining $17,000 was profit. The...
-
The example may be a sales tax or income tax proposal at any level of government. In your initial post, explain the revenue proposal, and evaluate it for equity, adequacy of revenue generation,...
-
How many 1,3-diaxial interactions occur in cis-decalin? In trans-decalin? Which compound has the lower energy and by how much?
-
Tell whether each of the following compounds is chiral. (a) Cis-1,2-dimethylcyclopropane (b) Trans-1,2-dimethylcyclopropane
-
When the United States Supreme Court denies certiorari for an appeal from a U.S. Circuit Court decision, this means that the justices agree with the decision of that circuit court and are giving it...
-
A servicing asset Multiple choice question. arises when a securitization entity collects cash from debtors and makes payments to investors. appears on the guarantor's balance sheet. arises when a...
-
firm's market value unchanged.So NPV uses a discount rate equal to the firm's cost of capital(or weighted average cost of capital).Decision Criteria:If NPV is greater than or equal to $0, accept the...
-
brfm17h_ch10.13m 1.Only Visited, Not Judged 2.Not Answered 3.Not Answered 4.Not Answered 5.Not Answered 6.Not Answered 7.Not Answered 8.Not Answered 9.Not Answered 10.Not Answered Question Workspace...
-
Lockheed Martin records inventories at the lower cost or the net realizable value. In the accounting report, the costs of a long-term contract represent the recoverable costs incurred for...
-
A full can of soda has a mass of 0.422 kg. It contains 3.59 x 104 m of liquid. Assuming that the soda has the same density as water, find the volume of aluminum used to make the can. Number 0.000236...
-
Compare the elasticity of the monopolistic competitors demand with that of a pure competitor and a pure monopolist. Assuming identical long-run costs compare graphically the prices and outputs that...
-
One of the significant and relevant accounts for this cycle is equipment. For this account, what would typically be the most relevant assertions for the auditor to consider? Why is it important for...
-
Allene, H2C = C = CH2, is somewhat unusual in that it has two adjacent double bonds. Draw a picture showing the orbitals involved in the and bonds of allene. Is the central atom sp2- or...
-
Allene (see problem 1.46) is related structurally to carbon dioxide, CO2. Draw a picture showing the orbitals involved in the and bonds CO2, and identify the likely hybridization of carbon.
-
Complete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the hybridization of the indicatedatoms. CH H3C. -- Caffeine 0= C
-
From 2006 to 2010, real gross domestic product (GDP) per capita in India grew an average of 7.11% per year. At that rate, according to the rule of 70, in approximately how many years will the size of...
-
The monthly US steel production market (in millions of tons per month) is described in the table below.An increase in the price of iron ore, a critical input in steel production, shifts the supply...
-
An employer faces a minimum wage control where it cannot pay its workers less than $10.25 per hour. The employer knows that workers value the jobs and are willing to work even for much less. The...

Study smarter with the SolutionInn App


