One of the following compounds has an intense yellow fluorescence when irradiated with UV light. Which one
Question:
One of the following compounds has an intense yellow fluorescence when irradiated with UV light. Which one do you think it is, and why?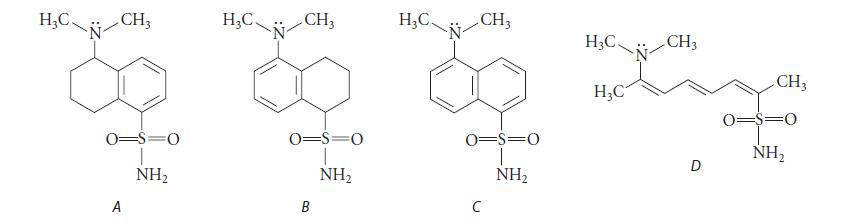
Transcribed Image Text:
H₂C. CH3 0=S=O A NH₂ H₂C. N CH₂ 0=S=0 NH₂ B H₂C. CH3 0=S=0 NH₂ C H3C. H₂C CH3 D CH3 0=$=0 NH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Based on the chemical structures of the four compounds shown in the image I think the compound that ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which one of the following compounds should have a * UV absorption at the greater max when the compound is dissolved in NaOH solution? Explain.
-
Refer to the Professional Judgment in Context feature at the beginning of the chapter. Additional details on PFG and Wasendorf are presented below. On July 14, 2012, Russell Wasendorf, Sr. attempted...
-
When a company has a winning product, it has it made. Or does it? Subaru is a winning company (one of the few automotive companies to sustain growth and profits in hard economic times) with various...
-
True or False? Azure storage is used by both Infrastructure as a Service ( ( laaS ) ) virtual machines, and Platform as a Service ( ( PaaS ) ) cloud services. True False
-
Why may net cash flow from operating activities on the cash flow statement be different from the amount of net income reported on the income statement?
-
David McClemore, the CFO of Ultra Bread, has decided to use an APT model to estimate the required return on the companys stock. The risk factors he plans to use are the risk premium on the stock...
-
Nereus Montemayor was an employee of VZ Hogs, a company that raises hogs and produces hog feed. VZ Hogs used an extruder manufactured by Sebright Products, Inc. to create hog feed out of discarded...
-
(Computation of Pension Expense, Amortization of Net Gain or Loss???Corridor Approach, Journal Entries for 3 Years) Hiatt Toothpaste Company initiates a defined-benefit pension plan for its 50...
-
Transform following Relational model into ER Mode id Author name dob AuthorContact id contact Manager writes authorid bookid Book bookid title edition libid memberld location opening Time...
-
What products are formed in the DielsAlder reactions of the following dienes and dienophiles? (a) (b) and HCO-C. and HC=C COCH3 COCH3
-
Refer to Fig. 15.11. At what pH does the fluorescence of fluorescein have the greater quantum yield, pH = 9 or pH = 5? How do you know? relative absorbance 300 488 nm pH=9 pH = 5 400 wavelength, nm...
-
Briefly explain the conditions when a contract modification shall be accounted for as a separate performance obligation.
-
How do control issues affect mergers?
-
Who are the major purchasers of regular preferred stock? How do tax considerations affect these purchases?
-
Formulate a problem statement and research objectives for the following situations: a. Six Flags Great America is deciding whether or not to open a new location in the Midwest. b. Buffalo Wild Wings...
-
What are some factors that acquiring firms consider when they set a bid price?
-
Describe the market multiple approach.
-
Courtney is an employee of Fremont Company. An average of three times a week, she works out during her lunch hour at a health club provided by Fremont. Discuss the taxability of Fremont's provision...
-
The age-old saying for investing is "buy low and sell high," but this is easier said than done. Investors who panic about falling prices sell their investments, which in turn lowers the price and...
-
The following compounds all show a single line in their 1H NMR spectra. List them in expected order of increasing chemical shift: CH4, CH2C12, cyclohexane, CH3COCH3, H2C = CH2, benzene
-
Predict the splitting pattern for each kind of hydrogen in the following molecules: (a) (CH3)3CH (b) CH3CH2CO2CH3 (c) trans-2-Butene
-
Predict the splitting pattern for each kind of hydrogen in isopropyl propanoate, CH3CH2CO2CH(CH3)2.
-
Write a C++ program (with two arrays) to: 1. Read up to 100 student numbers and test scores from the keyboard (provide a method to end the input before reaching 100 records); 2. Display the student...
-
What mechanisms underlie the process of desiccation in food preservation, and how does this method effectively inhibit microbial spoilage?
-
What is the process of filtration, and what are the underlying mechanisms that govern its functionality within various systems?

Study smarter with the SolutionInn App


