Refer to Fig. 15.11. At what pH does the fluorescence of fluorescein have the greater quantum yield,
Question:
Refer to Fig. 15.11. At what pH does the fluorescence of fluorescein have the greater quantum yield, pH = 9 or pH = 5? How do you know?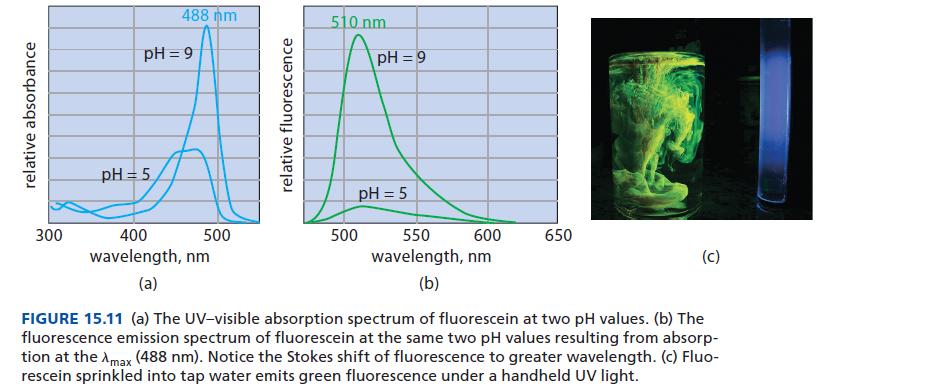
Transcribed Image Text:
relative absorbance 300 488 nm pH=9 pH = 5 400 wavelength, nm (a) 500 relative fluorescence 510 nm pH=9 pH = 5 500 600 550 wavelength, nm (b) 650 (C) FIGURE 15.11 (a) The UV-visible absorption spectrum of fluorescein at two pH values. (b) The fluorescence emission spectrum of fluorescein at the same two pH values resulting from absorp- tion at the Amax (488 nm). Notice the Stokes shift of fluorescence to greater wavelength. (c) Fluo- rescein sprinkled into tap water emits green fluorescence under a handheld UV light.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The figure presented shows the UVvisible absorption spectrum and the fluorescence emission spectrum ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
You will need to save money to reach your financial goals. To save money, you will need a plan to show what money comes in and what money goes out. This is known as a budget. You may already have a...
-
You are interviewing a candidate for a position at a call center. You need someone polite, courteous, patient, and dependable. The candidate you are talking to seems nice. But how do you know who is...
-
The following question was asked of executives: How do you know when to cut off research? The answers given: Thats a good question, a very good question, and some people dont know when to cut it off....
-
A tape player has three operations: play, fast forward and fast play. Play and fast forward are activated using the play and fast forward button respectively. These operations can be cancelled using...
-
When are expenses recognized under accrual accounting?
-
The Beta Batting Company manufactures wood baseball bats. Betas two primary products are a youth bat, designed for children and young teens, and an adult bat, designed for high school and college-...
-
On July 1, Chi Kong creates a petty cash fund with a balance of $300. During July, Elise Sautter, the fund custodian, signs the following petty cash tickets: Petty Cash Ticket Number 101 102 103...
-
A path cover of a directed graph G = (V, E) is a set P of vertex-disjoint paths such that every vertex in V is included in exactly one path in P. Paths may start and end anywhere, and they may be of...
-
The ABC Corporation has put together the following information regarding its two products: Blow dryer Leaf blower Direct Material$8$12 Direct Labor1015 Mfg. Overhead @$20 per mach. hr.2060 Cost from...
-
One of the following compounds has an intense yellow fluorescence when irradiated with UV light. Which one do you think it is, and why? HC. CH3 0=S=O A NH HC. N CH 0=S=0 NH B HC. CH3 0=S=0 NH C H3C....
-
Predict max for the UV absorption of each of the following compounds. (a) Et H T H H T H Et (b) H T H CH3
-
Let m denote the true average radioactivity level (picocuries per liter). The value 5 pCi/L is considered the dividing line between safe and unsafe water. Would you recommend testing H0: = 5 versus...
-
In Problem outline a procedure for solving the matrix game, then solve it. -2 1 -1 0 -1 3 2 4 1 -1 1 - 1 0 1 2
-
Given two values X and Y. You need to convert X to Y by performing various steps. In a step, we can either multiply 2 to the value or subtract 1 from it. You need to display the number of steps and...
-
The delay cost in dollars for the sell order of West Commerce shares is: A. \($1\),800. B. \($2\),000. C. \($2\),700. Michelle Wong is a portfolio manager at Star Wealth Management (SWM), an...
-
Briefly discuss any limitations associated with this research scenario and the specific design. Develop a hypothetical research scenario that would necessitate the use of a Solomon N-Group Design....
-
The implementation shortfall, in basis points (bps), for the sell order of West Commerce shares is closest to: A. 139. B. 198. C. 206. Michelle Wong is a portfolio manager at Star Wealth Management...
-
Why are direct materials price variances usually recorded at the time of purchase?
-
Provide an example of an aggressive accounting practice. Why is this practice aggressive?
-
The acid-catalyzed dehydration of 1-methylcyclohexanol yields a mixture of two alkenes. How could you use 1H NMR to help you decide which waswhich? CH CH - 0* CH2
-
How could you use 1H NMR to distinguish between the following pairs ofisomers? (a) CH3CH=CHCH2CH3 and CH2 H H2H () CH20CH2CH and CH3OCH2CH2CH3 (c) CHC,H and CH3CH2CH3 (d) HCICH)H and CH3CH=CHCH3
-
Propose structures for compounds with the following formulas that show only one peak in their 1H NMR spectra: (a) C5H12 (b) C5H10 (c) C4H8O2
-
What is Preferred Stock? Discuss the characteristics of Preferred Stock over Common Stock. MINIMUM ONE PAGE REQUIREMENT. 2. What are Stock Options? Discuss the TWO primary reasons why corporations...
-
If your organization was given $10,000 for digital marketing for a quarter (3 months), how would you spend it?
-
For the following function, list the intervals where f(x) is concave up and where it is concave down. Find any points of inflection. Do NOT use a graph to answer the question. f(x)=3x+2x-12x+5

Study smarter with the SolutionInn App


