Outline a synthesis of each of the following compounds from the indicated starting material. Begin each synthesis
Question:
Outline a synthesis of each of the following compounds from the indicated starting material. Begin each synthesis with a retrosynthetic analysis.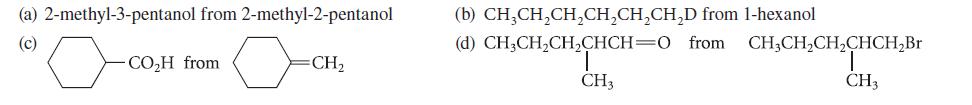
Transcribed Image Text:
(a) 2-methyl-3-pentanol from 2-methyl-2-pentanol (c) -CO₂H from =CH₂ (b) CH3CH₂CH₂CH₂CH₂CH₂D from 1-hexanol (d) CH3CH₂CH₂CHCH=0 from. CH3CH₂CH₂CHCH₂Br CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a 2Methyl3pentanol can be prepared by hydroborationoxidation of 2methyl2pentene which ca...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline a synthesis for each of the following compounds from the indicated starting material and any other reagents. (a) l-chloro-3, 5-dinitrobenzene from benzene (b) 2-chloro-4,6-dinitrophenol from...
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) Benzyl methyl ether from toluene (b) (c) (d) CH2CH O from cyclopentene 7-CO.H...
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) 1-cyclohexyl-2-methyl-2-prupanol from bromocyclohexane (b) PhNHCH2CH2CH(CH3)2...
-
The Chief Financial Officer at Ford Motor Company is said to usea hybrid-costing system. Define the hybrid-costing system. Explainthe advantages to this company to use this system. I want a 10 page 2...
-
Refer to the data in PE 6-3. Assume that all of the 80 books sold on credit were sold to a single customer and that the terms of the credit sale were 2/10, n/30. Make the journal entry necessary to...
-
The balance sheets of Barrier Corporation as of December 31, Year 2, and Year 1, and its statement of income and retained earnings for the year ended December 31, Year 2, follow: BARRIER CORPORATION...
-
Are there any circumstances when top management will be interested in detailed ecological accounting information? Explain.
-
Stewart Construction Corporation had a net income of $16,500 during 2014. In that year, the company had depreciation expense of $7,000. Accounts Receivable increased by $5,500 and Accounts Payable...
-
List five with an explanation of the obligatory conditions of Zakat to be eligible as alms asset?
-
The primary alcohol 2-methoxyethanol, CH 3 OCH 2 CH 2 OH, can be oxidized to the corresponding carboxylic acid with aqueous nitric acid (HNO 3 ). The by-product of the oxidation is nitric oxide, NO....
-
The rates of the reactions in Eqs. 10.71ab are increased when the thiol is ionized by a base such as sodium ethoxide. Suggest a mechanism for Eq. 10.71a that is consistent with this observation, and...
-
Graphs of populations of two species are shown. Use them to sketch the corresponding phase trajectory. species l 200 species 2 150 100 50
-
An article in the Wall Street Journal in early 2023 discussed Teslas decision to cut the prices of the electric vehicles it sells in China. According to the article, The company said it is responding...
-
In 2022 and 2023, states including Pennsylvania, Utah, and Maryland removed their requirement that those applying for some state government jobs have college degrees. In the case of Pennsylvania, 92...
-
Employers who offer health insurance typically allow existing employees to enroll or change their coverage just once a year during open enrollment in the fall. Why might employers not allow their...
-
An article in the New York Times noted that at the time he took office, President Biden supported the idea of a public option: a government-run health plan that all Americans could choose to buy...
-
An article in the Wall Street Journal reports that some hospitals charge up to 10 times as much as others for standard medical scans. The article also notes: Health economists say the disparities...
-
Refer to the information from Problem 6.47. Physical Units Beginning WIP (25% complete) ....... 11,000 Started during January ........... 74, 0000 Total to account for ............ 85,000 Good units...
-
Feller Company purchased a site for a limestone quarry for $100,000 on January 2, 2019. It estimate that the quarry will yield 400,000 tons of limestone. It estimates that its retirement obligation...
-
What stereochemistryantarafacial or suprafacialwould you expect to observe in the following reactions? (a) A photo chemical [1, 5] sigma tropic rearrangement (b) A thermal [4 + 6] cyclo addition (c)...
-
The following thermal isomerization occurs tinder relatively mild conditions. Identify the pericyclic reactions involved, and show how the rearrangementoccurs. CH C- CeH5. C&H5 "CH CD CoH5 CgH5 CEH5...
-
Would you expect the following reaction to proceed in a conrotatory or disrotatory manner? Show the stereochemistry of the Cyclobutene product, and explain youranswer. hv
-
An electron undergoes a transition from the 3rd energy level to the 1st energy level in a hydrogen atom. The wavelength of the radiation associated with this transition is a.bc x 10-d m. Record abcd....
-
You receive a call from Lydia Asoma of Acme. She says she is the HR Manager at Acme and wants you to act for her as her solicitor to defend allegations being made against her by Acme concerning the...
-
Consider the same mechanism again, with m=2.5 kg , d=585 mm , k=200 N/m , and s=1.72 . What is the maximum angular velocity allowable if the collar is to remain at a constant distance r=2600 mm from...

Study smarter with the SolutionInn App


