Outline a synthesis of m-nitroacetophenone from benzene; explain your reasoning. ON. C-CH3 m-nitroacetophenone
Question:
Outline a synthesis of m-nitroacetophenone from benzene; explain your reasoning.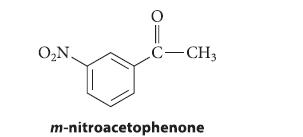
Transcribed Image Text:
O₂N. C-CH3 m-nitroacetophenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Because both groups are metadirecting groups it might seem that either c...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline a synthesis of 1-phenylcyclohexene from benzene and cyclohexene.
-
Outline a synthesis of 1-phenylcyclohexene from benzene and cyclohexene.
-
Starting with benzene or toluene, outline a synthesis of each of the following compounds using diazonium salts as intermediates. (You need not repeat syntheses carried out in earlier parts of this...
-
Suppose that Marthas income rises to $42,000 per year, and that she increases her consumption of health care visits by fi ve visits. Using the graphs for Exercise 1, draw the new equilibrium. What is...
-
Interpreting common-size income statements Exhibit 4.7 presents common-size income statements for Standard Denim and Blue Label Jeans, two apparel retailing firms, for three recent years. In addition...
-
Refer to the CVS annual report in the Supplement to Chapter 5 and to the following data (in millions) for Walgreens: net sales, $59,034.0 and $53,762.0 for 2008 and 2007, respectively, accounts...
-
A force of magnitude \(F\) is exerted on the leftmost face of two blocks sitting next to each other on a slippery surface, with two of their faces touching. The inertia of the block on the right is...
-
Three years ago, the Oak Street TCBY bought a frozen yogurt machine for $11,200. A salesman has just suggested to the TCBY manager that she replace the machine with a new, $13,500 machine. The...
-
Morning Smiles Coffee Company manufactures Stoneware French Press coffee makers. During the month of March, 8,100 coffee makers were completed at a cost of goods manufactured of $607,500. Suppose...
-
Predict the predominant product(s) from: (a) Monosulfonation of m-bromotoluene (b) Mononitration of m-bromoiodobenzene
-
The iodination reaction discussed in this section can be carried out on the amino acid tyrosine and related compounds in the laboratory with iodide ion in the presence of the enzyme thyroid oxidase....
-
Use the unit circle to complete Exercises (see Example 5).Find sin 0. Data from Example 5 (a) Use the unit circle to find cos 90 and sin 90. A 90 angle, when placed in standard position, intersects...
-
Why is it important to go through the process of closing, even for projects that are terminated early?
-
Should activities on the critical path be outsourced? Why or why not?
-
If your sponsor asks for an estimate as to how much more money your team needs to complete all project work as scheduled, which of the following formulas might you use? a. BAC 2 EAC b. EV/PV c. (BAC...
-
What does a typical closeout report include?
-
What marks the transition between the planning and executing project phases?
-
The following data come from independent samples drawn from normally distributed populations. Use these data to test the following hypotheses. Let the Type I error rate be .05. H0: 1 2 = 0 Ha: 1 2...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
Show two different Friedel-Crafts acylation reactions that can be used to prepare the following compound. CH3 CH CH3
-
Draw the structure of each of the following compounds. (a) m-nitrotoluene (b) 1-bromo.2-propylbenzene (c) Benzyl methyl ether (d) o-cresol
-
Using the information in Table 16.2, predict the product(s) of Table 16.2 Friedel-Crafts alkylation of a large excess of ethylbenzene with chloromethane in the presence of A1C13. Eq. 16.23 TABLE 16.2...
-
A plate carries a charge of-2 C, while a rod carries a charge of 2.50 C. How many electrons must be transferred from the plate to the rod, so that both objects have the same charge? N-Number...
-
A cube of wood having an edge dimension of 19.9 cm and a density of 653 kg/m floats on water. (a) What is the distance from the horizontal top surface of the cube to the water level? cm (b) What mass...
-
A siphon is used to drain water from a tank as illustrated below. Assume steady flow without friction. (a) If h = 1.27 m, find the speed of outflow at the end of the siphon. m/s (b) What is the...

Study smarter with the SolutionInn App


