Potassium tri(isopropoxy)borohydride sometimes finds use as a source of nucleophilic H: (hydride ion). Suggest a mechanism for
Question:
Potassium tri(isopropoxy)borohydride sometimes finds use as a source of nucleophilic H:– (hydride ion).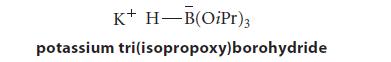
Suggest a mechanism for the substitution reaction in Fig. P18.91 that accounts for the stereochemistry of the reaction.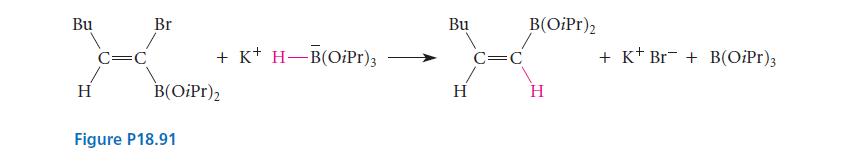
Transcribed Image Text:
K+ H-B(OiPr)3 potassium tri(isopropoxy)borohydride
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The hydride of potassium triisopropoxyborohydride forms a Lewis acidbase associ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
We have seen that NaH is a strong base but a weak nucleophile. In contrast, lithium aluminum hydride (LAH) is a reagent that can serve as a source of nucleophilic hydride ion: In this case, LAH...
-
The reaction between potassium superoxide, KO2, and CO2, 4 KO2 + 2 CO2 2K2CO3 + 3 O2 is used as a source of O2 and absorber of CO2 in self-contained breathing equipment used by rescue workers. (a)...
-
The rate law for the substitution reaction of 2-bromobutane and HO- in 75% ethanol and 25% water at 30C is rate = 3.20 10-5[2-bromobutane][HO-] + 1.5 10-6[2-bromobutane] What percentage of the...
-
The Holtz Corporation acquired 80 percent of the 100,000 outstanding voting shares of Devine, Inc., for $7.20 per share on January 1, 2014. The remaining 20 percent of Devines shares also traded...
-
The adjusted trial balances of Superior International, Inc., at August 31, 2012, and August 31, 2011, include the following amounts: Analysis of the accounts at August 31, 2012, reveals the following...
-
Motorists arrive at a Gulf station at the rate of two per minute during morning hours. Assume that the arrival of motorists at the station follows a Poisson distribution. In addition to providing the...
-
What is the internal rate of return of the following cash flow diagram? a. 20 percent b. 18.2 percent c. 17.5 percent d. 15 percent $30 $31 0 1 2 3 $30 $15
-
Selected comparative financial statements of Korbin Company follow. Required 1. Compute each years current ratio. (Round ratio amounts to one decimal.) 2. Express the income statement data in common-...
-
a) Your roommate heard that you took Fundamentals of Finance with Prof. Hasler and that you learnt about the idea of building a nest egg for your retirement. Your roommate has a crazy idea and asks...
-
Citalopram is used as an antidepressant. A biologist, Heywood U. Clonum, has argued that this compound could be hazardous because it could release toxic cyanide ( C N) and fluoride ions by...
-
Bombykol is the mating pheromone of the female silkworm moth. Using the result in Problem 18.89, propose a synthesis of bombykol, using both HC C(CH 2 ) 9 OH (10-undecyne-1-ol) and 1-pentyne as...
-
Using the state pharmacy laws and rules from your state, does your state have any drug repository or take-back programs? If yes, describe the details.
-
What makes the challenge meaningful for those who push their own limits to get extraordinary things done, who risk their security to change the social condition or risk their lives to save others or...
-
How can technology and several popular search engines filter your information making your searches more "funneled" and "narrow our view." Describe why and how this happens.
-
Reflect on your current understanding of academic and professional integrity. Using at least 300 words, explain how you would respond in the given situation. Anika has been tasked to submit a report...
-
Carefully review all the creative and media tactics used in the campaign. What the Lionsgate do well and what could have been done better? should be strictly based on hunger games catching fire by...
-
What does author Allan Johnson mean when he says that, "Oppression and dominance name social realities that we can participate in without being oppressive or dominating people."? Why is it so...
-
TeleTech Corporation manufactures two different fax machines for the business market. Cost estimates for the two models for the current year are as follows: The firms management expects to operate at...
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
In contrast to the reaction with dilute alkali (Section 18.6), when concentrated solutions of NaOH are used, acetoacetic esters undergo cleavage as shown below.
-
(a) What product would you expect from a Dieckmann condensation of diethyl heptanedioate? (b) Can you account for the fact that diethyl pentanedioate (diethyl glutarate) does not undergo a Dieckmann...
-
Write a detailed mechanism for the following reaction. OEt NaOEt EtO OEt OEtEtO Ethyl crotonate Diethyl oxalate
-
Write a C program that allows a single Player (the user) to play a simple three dice game of chance against "The Odds". 1. There is a single player, with three eight sided dice. 2. The sides of each...
-
6. (15 pt) Function recur() has its prototype shown below Complete C function recur() corresponding to the IA32 code on the right. int recur(int a[], int n) { recur: pushl %ebp movl %esp, %ebp pushl...
-
2. Consider the following program: def my decorator (func_to_decorate): def my wrapper (*args, **kwargs): print ("In wrapper, wrapping ()* .format(func_to_decorate_name)) result func_to_decorate...

Study smarter with the SolutionInn App


