Predict the NMR spectrum, including approximate chemical shifts, of the following compound. Explain your reasoning. OCH3 CH3O-CH-C-CH-OCH3
Question:
Predict the NMR spectrum, including approximate chemical shifts, of the following compound. Explain your reasoning.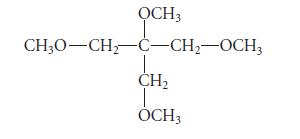
Transcribed Image Text:
OCH3 CH3O-CH₂-C-CH₂-OCH3 CH₂ OCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The spectrum will contain three resonances which arise fr...View the full answer

Answered By

Gaurav Soni
Teaching was always an area where I can pursue my passion. I used to teach my friends and junior during my school and college life. After completing my professional qualification (chartered accountancy) and before joining my job, I also joined an organization for teaching and guidance to my juniors. I had also written some articles during my internship which later got published. apart from that, I have also given some presentations on certain amendments/complex issues in various forms.
Linkedin profile link:
https://www.linkedin.com/in/gaurav-soni-38067110a
5.00+
7+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the approximate chemical shifts of the protons in the following compounds. (a) Benzene (b) Cyclohexane (c) CH3-O-CH2CH2CH2Cl (d) CH3CH2-C¡¡C-H (e) (f) (CH3)2CH-O-CH2CH2OH (g) (h)...
-
Predict the chemical shifts for the signals in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) (d) (e)
-
The proton NMR spectrum of 2-pyridone gives the chemical shifts shown. (a) Is 2-pyridone aromatic? (b) Use resonance forms to explain your answer to (a). Also explain why the protons at (7.31and...
-
What was the action you are most proud of, or that is the most meaningful to you? What was the civic issue that was involved in your action? When and how did you first hear about that issue? Why did...
-
How is an NSF check accounted for in the accounting records?
-
ABC Company and XYZ Company need to raise funds to pay for capital improvements at their manufacturing plants. ABC Company is a well-established firm with an excellent credit rating in the debt...
-
George Oppenheimer, an agent for Wellington Farms of Massachusetts, Inc., had contacted Mark Kiriakou from the Capital Area Food Bank regarding an order for frozen turkey meat. In an exchange of...
-
Borders Group, Inc., presented this information in its 10-Ks: Required a. Compute the following liquidity ratios for 2009 and 2008: 1. Days sales in inventory 2. Inventory turnover (use ending...
-
What are the reasons to use a relational database? What data does not fit well in the relational model?
-
Propose a structure for a compound with the formula C 7 H 14 with the NMR spectrum shown in Fig. 13.18. Explain in detail how you arrived at your structure. 2400 absorption Lii 8 2100 7 1800 CH14 6...
-
(a) Consider entries 1 through 4 of Table 13.1. How does the chemical shift of a proton vary with the electronegativity of the neighboring halogen? (b) Compare entries 2, 5, and 6 of Table 13.1. How...
-
Victor sells to Bonnie a refrigerator for $600 payable in monthly installments of $30 for twenty months. Bonnie signs a security agreement granting Victor a security interest in the refrigerator. The...
-
How are incidence rates calculated? What do the results indicate?
-
What are the roles and responsibilities of an effective and active Board of Directors?
-
How should a board of directors be involved in the executive leadership of an organization?
-
What would be effective ways to recruit qualified professionals to work for Tommy?
-
Training programs are frequently the first items eliminated when management wants to cut costs. Why do you believe this occurs?
-
Capital gains of individuals are taxed at a 15% rate (zero% for 10% or 15% marginal tax rate taxpayers). Capital gains of corporations are taxed at the corporations marginal tax rate. In reducing the...
-
As of January 1, 2018, Room Designs, Inc. had a balance of $9,900 in Cash, $3,500 in Common Stock, and $6,400 in Retained Earnings. These were the only accounts with balances in the ledger on January...
-
Pyridine is a flat, hexagonal molecule with bond angles of 120?. It undergoes substitution rather than addition and generally behaves like benzene. Draw a picture of the ? orbital?s of pyridine to...
-
To be aromatic, a molecule must have 4n + 2? electrons and must have cyclic conjugation. 1, 3, 5, 7, 9-Cyclodecapentaene fulfills one of these criteria but not the other and has resisted all attempts...
-
Draw the five resonance structures of the cyclopentadienyl anion. Are all carbon carbon bonds equivalent? How many absorption lines would you expect to see in the 1H NMR and 13C NMR spectra of the...
-
Elizabeth and William have a daughter, age 11, and have spent $8,480 on qualified childcare so both Elizabeth and William can work. The couple's combined AGI is $112,600 with William earning $43,800...
-
Given the following graph, what is the normalized graph cut measure when the nodes are grouped into A (blue) and B (green) as shown below. A 2 2 2 2 2 4 3 1 1 2 2 B 2 2 22 2 2 3
-
Gital inherited a piece of land when a close relative died on July 26, 2020. The FMV of the land at the date of death was $40,000 and cost the relative $20,000 in 2014. Gital sold the land on March...

Study smarter with the SolutionInn App


