Predict the chemical shifts for the signals in the 1 H NMR spectrum of each of the
Question:
(a)
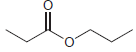
(b)
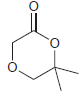
(c)
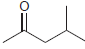
(d)
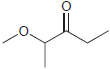
(e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a b c d e All four methylene groups are equivalent so the compound will have only one signal in ...View the full answer

Answered By

Zablon Gicharu
I am an educator who possesses the requisite skills and knowledge due to interacting with students for an extended period. I provide solutions to various problems in step-by-step explanations, a well-thought approach and an understandable breakdown. My goal is to impart more straightforward methodologies and understanding to students for more remarkable achievements.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the chemical shifts of the protons in the following compounds. (a) (b) (c) (d) (e) (f) HC(CH3)3 CH CH CH3 , - CH,CCCH OH CH,CH,-C-OH CH3 CHC-CH Br
-
Predict the approximate chemical shifts for the different hydrogen's in thesecompounds: CI CI a) CH,CH,CH3 b) CH;CHCH3 c) CH,COCH,CH3 d) CH;CHCH2
-
Explain the direction of the chemical shifts for Fe(0), Fe(II), and Fe(III) in Figure 22.20. Fe 2p at normal emission Fe 2p32 Fe 2p12 Fe (0) Fe/MgO(001) Fe(0) Fe;OM9O(001) Fe(ll & II) Fe(ll) Fe(lI)...
-
Net sales revenue, net income, and common stockholders' equity for Vallen Optical Corporation, a manufacturer of contact lenses, follow for a four-year period. Requirements 1. Compute trend...
-
What is a "liquidity premium?" When do investors increase their attention to such a premium?
-
Between the following two firms, which firm is more likely to be successful in exploiting its sources of sustained competitive advantage in its home market in a highly competitive, non-domestic...
-
A four cylinder, four stroke petrol engine is to be design to give brake power of \(185 \mathrm{~kW}\) at 35 RPS. The stroke bore ratio is to be 1.5. Assuming mechanical efficiency of \(75 \%\) and...
-
1. What type of installation did Hudson Banc use for its new system? Was it an appropriate choice? 2. How could the operational problems have been avoided? Two regional banks with similar geographic...
-
You have decided to go into the business of selling Beamers. You decide to operate the business as a corporation, Chris's Beamer Biz, Inc. On January 1, 20X1 the company issued you 100 shares of...
-
The position of a particle as a function of time is given by r(vector) = (5.0i + 4.0j)t 2 m, where t is in seconds. a. What is the particles distance from the origin at t = 0, 2, and 5 s? b. Find an...
-
Identify the structure of a compound with molecular formula C 9 H 20 that exhibits four CH 2 groups, all of which are chemically equivalent. How many total signals would you expect in the 1 H NMR...
-
A 1 H NMR spectrum was acquired for each of the following constitutional isomers. Comparison of the spectra reveals that only one of these spectra exhibits a signal between 6 and 7 ppm. Identify the...
-
At one point in his study of a falling body starting from rest, Galileo conjectured that its velocity at any time is proportional to the distance it has dropped. Using this hypothesis, set up the...
-
What is a rights offering?
-
Why might PESTLE analysis be particularly appropriate for companies in the oil industry?
-
Explain how the value net and complementors help organizations understand how to add value for customers.
-
What is a tax-exempt lease?
-
Describe the theory of the business on which Blockbuster and Netflix was based. Explain how this resulted in Blockbuster going bankrupt and Netflix dominating film and TV streaming.
-
When calculating taxable income, partnerships are not allowed which of the following deductions? a. Miscellaneous expenses. b. Qualified business income deduction. c. Depreciation. d. Cost of goods...
-
A superior criticized a sales manager for selling high-revenue, low-profit items instead of lower-revenue but higher-profit items. The sales manager responded, My income is based on commissions that...
-
Name the five isomers of C6H14. Discuss.
-
Explain why each of the following names is incorrect: (a) 2, 2-Dirnethyl-6-ethytheptane (b) 4-Ethyl-5, 5-dirnethylpentane (c) 3.Ethyl-4, 4-dimcthylhexane (d) 5, 5, 6-Trimcthyloctane (e)...
-
Propose structures and give IUPAC names for the following: (a) A diethyldimethyihexane (b) A (3-methylbutyl)-substituted alkane
-
Describe, in steps, at least one strategy used to attack embedded operating systems. Describe, in steps, at least one strategy used to defend against the chosen attack.
-
how does a hypercompetetice environment challenge those in an industry
-
Describe how managers can reduce unnecessary costs, including real - world examples of how it has been done

Study smarter with the SolutionInn App


