Propose a structure for the compound that has the following spectra: Mass spectrum: m/z = 152, 150
Question:
Propose a structure for the compound that has the following spectra: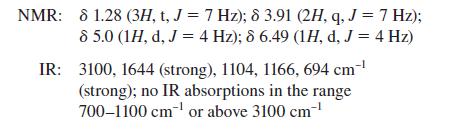
Mass spectrum: m/z = 152, 150 (equal intensity; double molecular ion)
Transcribed Image Text:
NMR: 81.28 (3H, t, J = 7 Hz); 8 3.91 (2H, q, J = 7 Hz); 8 5.0 (1H, d, J = 4 Hz); 8 6.49 (1H, d, J = 4 Hz) IR: 3100, 1644 (strong), 1104, 1166, 694 cm-¹ (strong); no IR absorptions in the range 700-1100 cm¹ or above 3100 cm-¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The mass spectrum indicates that the compound contains a single bromine double molecular ...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C4H8O has a strong IR absorption at 1730 cm-1. Its mass spectrum includes key peaks at m/z 44 (the base peak) and m/z 29. Propose a structure for the compound and...
-
An unknown compound gives a molecular ion of m / z70 in the mass spectrum. It reacts with semicarbazide hydrochloride to give a crystalline derivative, but it gives a negative Tollens test. The NMR...
-
The IR, NMR, and mass spectra are provided for an organic compound. (a) Consider each spectrum individually, and tell what characteristics of the molecule are apparent from that spectrum. (b) Propose...
-
How to find the expected utility of profit under each alternative crop Table 1. Annual Returns to Cropping Alternatives ($ profit / acre) Outcome Worst Bad OK Good Great Probability 0.1 0.2 0.4 0.2...
-
Dick Haney is opening a new business that will sell sporting goods. It will initially be a small operation, and he is concerned about the security of his assets. He will not be able to be at the...
-
What is the NAL for Wildcat? What is the maximum lease payment that would be acceptable to the company? The Wildcat Oil Company is trying to decide whether to lease or buy a new computer-assisted...
-
Plaintiff contracted to install a boiler for defendant. After plaintiff had installed and tested the boiler, but before final payment to plaintiff had been made, defendant took custody of the boiler...
-
Dr. Massy, who specializes in internal medicine, wants to analyze his sales mix to find out how the time of his physician assistant, Consuela Ortiz, can be used to generate the highest operating...
-
Block 1 m m Block 2 Note: Figure not drawn to scale. Two blocks are connected by a string of negligible mass that passes over massless pulleys that turn with negligible friction, as shown in the...
-
The NMR spectrum of vitamin D 3 is given in Fig. P13.58. Interpret the resonances marked with asterisks (*) by indicating the part of the structure to which they correspond. (Do not try to assign the...
-
The two protons H a and H b in 1,2,3-trichloropropane have slightly different chemical shifts, and the splitting pattern of each is a doublet of doublets. For one proton, J 5 9.0 Hz and 4.9 Hz; for...
-
In Exercises write an equation for the line through P that is (a) Parallel to L, (b) Perpendicular to L P(-2, 2), L: 2x + y = 4
-
BlackRock, Inc. (BLK) has a capital structure that consists of common stock equity and debt. The market capitalization of its equity is $77.004 billion and its debt has a market value of $4,967...
-
Hugh, an investment analyst in Brisbane, is analyzing three separate convertible bonds issued by Woolworths Limited, the Commonwealth Bank of Australia, and BHP Billiton. For each of them, calculate...
-
If elasticity of demand is 2 and price is raised from $10 to $11, by what percentage will quantity demanded fall?
-
If elasticity of demand is 0.5 and price is lowered from $20 to $19, by what percent age will quantity demanded rise?
-
A bond that matures in 8 years has a par value of $1,000 and an annual coupon payment of $70; its market interest rate is 9%. What is its price?
-
Explain the circumstances under which a scholarship would not be excluded from gross income.
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
Draw all isomers of C4H9Br, name them, and arrange them in order of decreasing reactivity in the SN2 reaction.
-
The following Walden cycle has been carried out. Explain the results, and indicate where Walden inversion isoccurring. OTos H-CH TosCI CHH2 eat CHH2 CHH2 CHCH2 lalp = +33.0 lalp = +31.1 [alp = -19.9...
-
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actualproduct. OCCH3)3 . H2CH Br (a) CHCH-CH (CH]l3c (b) Na* - CI LCH3 LCH3 (c) Socil,...
-
Find the weighted mean of the hourly salaries: 11 employees have an hourly salary of 22 dollars, 16 employees have an hourly salary of 77 dollars, 13 employees have an hourly salary of 63 dollars, 13...
-
A hard Drive manufacturers is required to ensure that the meantime between failures for its new hard drive is 1 million hours. Astress test is designed that can simulate the workload at a muchfaster...
-
A function and its graph are given. Use the graph to find the vertical asymptotes, if they exist. Confirm your results analytically. f(x)= 72 x+2 -15 10 x

Study smarter with the SolutionInn App


