The NMR spectrum of vitamin D 3 is given in Fig. P13.58. Interpret the resonances marked with
Question:
The NMR spectrum of vitamin D3 is given in Fig. P13.58.
Interpret the resonances marked with asterisks (*) by indicating the part of the structure to which they correspond.
(Do not try to assign the individual resonances within the groups.) Explain your choices.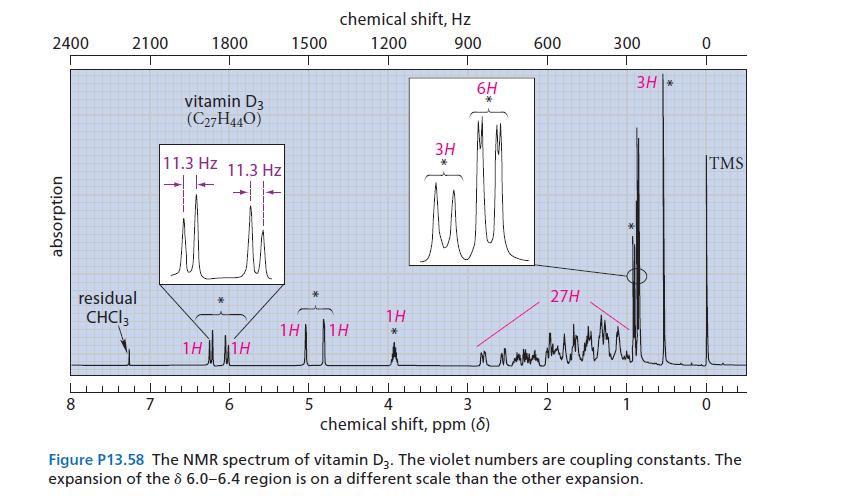
Transcribed Image Text:
HO CH₂H H H₂C H₂C- H H vitamin D3 CH3 CH₂CH₂CH₂CH T CH 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The relevant protons a...View the full answer

Answered By

Mehwish Aziz
What I have learnt in my 8 years experience of tutoring is that you really need to have a friendly relationship with your students so they can come to you with their queries without any hesitation. I am quite hardworking and I have strong work ethics. Since I had never been one of those who always top in the class and always get A* no matter what, I can understand the fear of failure and can relate with my students at so many levels. I had always been one of those who had to work really hard to get decent grades. I am forever grateful to some of the amazing teachers that I have had who made learning one, and owing to whom I was able to get some extraordinary grades and get into one of the most prestigious universities of the country. Inspired by those same teachers, I am to be like one of them - who never gives up on her students and always believe in them!
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The NMR spectrum of toluene (methylbenzene) was shown in Figure 13-11. (a) How many different kinds of protons are there in toluene? (b) Explain why the aromatic region around 7.2 is broad, with...
-
The NMR spectrum of cinnamaldehyde follows. (a) Determine the chemical shifts of Ha, Hb and Hc. The absorption of one of these protons is difficult to see; look carefully at the integrals. (b)...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
2) Assume that your widget manufacturing company has a total annual demand of N widgets per year evenly distributed across the year. Each widget cost $b dollars in material and manufacturing costs to...
-
The bank statement of Gear Supplies included a $300 NSF check that one of Gear's customers had written to pay for services that were provided by Gear. Required a. Show the effects of recognizing the...
-
Quartz Corporation is a relatively new firm. Quartz has experienced enough losses during its early years to provide it with at least eight years of tax loss carryforwards. Thus, Quartzs effective tax...
-
Abbott Industries is a well-known supplier of pharmaceuticals worldwide. Founded by Dr. Wallace Abbott, the company was incorporated in 1900 after he had been developing and making pharmaceuticals...
-
You are provided with the following information for Matthew Inc. for the month ended October 31, 2014. Matthew uses a periodic method for inventory. Instructions (a) Calculate (i) ending inventory ,...
-
A spherical beach pebble is completely submerged under sea water of mass density (not weight density!) 1041 kg/m3. The water exerts an upward buoyant force of magnitude 181 N on the pebble. What is...
-
You work for a reputable chemical supply house. An angry customer, Fly Ofterhandle, has called, alleging that a sample of 2,5-hexanediol he purchased cannot be the correct compound. As evidence, he...
-
Propose a structure for the compound that has the following spectra: Mass spectrum: m/z = 152, 150 (equal intensity; double molecular ion) NMR: 81.28 (3H, t, J = 7 Hz); 8 3.91 (2H, q, J = 7 Hz); 8...
-
Plaintiff applied for a job at Neiman Marcuss Oak Brook, Illinois, store as an entry-level dress collections sales associate. Ohle was interviewed and was informed that she should expect an offer for...
-
If the price of iPods is reduced by 50 percent and the quantity of songs demanded on iTunes rises by 25 percent, then the cross elasticity of demand for iPods and iTune songs is ______. a) 5.0 b) 1.0...
-
Let \(X\) be a random variable representing the minimum of the two numbers of dots that are facing up after a pair of fair dice is rolled. Define the appropriate probability density for \(X\). What...
-
Suppose Table 1 shows your demand schedule for cans of soda. (a) What is your total utility from three cans of soda? (b) What is your marginal utility from the third can of soda? (c) If price were...
-
Suppose Table 2 shows your demand schedule for CDs. (a) What is your total utility from four CDs? (b) What is your marginal utility from the fourth CD? (c) If the price is $2, how much will your...
-
What is the lowest price the firm would accept in the long run? Cost ($) 130 120 110 100 90 80 70 60 50 40 Figure 3 MC ATC AVC 24 6 8 10 12 14 16 18 20 22 Output
-
What tax relief is provided to U.S. citizens who earn income in a foreign country and pay taxes in that country?
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
What products would you expect from the reaction of 1-bromopropane with each of the following? (a) NaNH2 (b) KOC (CH3)3 (c) NaI (d) NaCN (e) NaC CH (f) Mg, then H2O
-
Which reactant in each of the following pairs is more nucleophilic? Explain. (a) NH2 or NH3 (b) H2O or CH3CO2 (c) BF3 or F (d) (CH3)3P or (CH3)3N (e) I or Cl (f) C N or OCH3
-
Propose structures for compounds that fit the following descriptions: (a) An alkyl halide that gives a mixture of three alkenes on E2 reaction (b) An organo halide that will not undergo nucleophilic...
-
Esperanza quit her job paying her $78,000 per year to operate a food truck firm selling poke bowls. In her first year of operation, she: spent $58,000 for ingredients; rented a truck for $28,000;...
-
What is the strictest condition that must be met before recognizing revenue from a donor with restrictions?
-
Canadian Armed Forces (CAF) members deployed to certain locations are entitled to additional compensation in recognition of the dangers and conditions associated with each specific location. What is...

Study smarter with the SolutionInn App


