Show a curved-arrow mechanism for the first step, and the structure of the cyclic intermediate formed, when
Question:
Show a curved-arrow mechanism for the first step, and the structure of the cyclic intermediate formed, when an alkene is treated with KMnO4. A Lewis structure for the permanganate ion is as follows: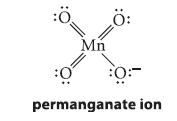
Transcribed Image Text:
:Ö Mn Ö: :0: permanganate ion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
This mechanism is much like the one shown for ...View the full answer

Answered By

Deborah Joseph
My experience has a tutor has helped me with learning and relearning. You learn everyday actually and there are changes that are made to the curriculum every time so being a tutor has helped in keeping me updated about the present curriculum and all.
I have also been able to help over 100 students achieve better grades particularly in the categories of Math and Biology both in their internal examinations and external examinations.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An alkene is treated with OsO4 followed by H2O2. When the resulting diol is treated with HIO4, the only product obtained is an unsubstituted cyclic ketone with molecular formula C6H10O. What is the...
-
Bank Corp has a defined benefit plan with 90 employees. 10 of the employees belong to a union. 20 of the employees have just started working within the last nine months. What is the minimum number of...
-
A firm is expected to pay a dividend of $3.90 one year from now and $4.25 two years from now and $4.30 three years from now. The firm's stock price is expected to be $110.50 in four years. What is...
-
The figure below represents a schematic of pipe network. A rate of 35 Ls, is pumped to feed two lines (3-4-5-6; and 2-7-8). The length and diameter of each pipe segment are listed in the table....
-
Company G received a bank statement at the end of the month. The statement contained the following: Ending balance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Compare enterprise computing systems to transaction processing systems. Provide three examples of each type of system.
-
Apply the marketing-myopia concept to print media, magazines, and newspapers. What is the implication?
-
McEntire Corporation began operations on January 1, 2009. During its first 3 years of operations, McEntire reported net income and declared dividends as follows. The following information relates to...
-
Water bottle in a hot car. In the American Southwest, the temperature in a closed car parked in sunlight during the summer can be high enough to burn flesh. Suppose a bottle of water at a...
-
Give the product(s) expected when each of the following compounds is treated with periodic acid. OH OH | -CH-CH3 (b) | PhCHCHCHOH OH
-
From what alkene could each of the following glycols be prepared by the OsO 4 or KMnO 4 method? OH I CH3CHOCHCHCHCHOH (b) OH -CHOH (c) meso-4,5-octanediol (d) ()-4,5-octanediol
-
Explain the potential impact of national image and counterfeit products on international product strategy.
-
A soft drink in a cup can be cooled without dilution using chilled plastic spheres that are 1.0 cm in diameter instead of ice. Assume that 300 mL of the spheres initially at 20C are added to 300 mL...
-
1. Give some examples of technology-based regulation. 2. Give some examples to show how safety-based legislation can be inefficient. 3. Give some examples to show how efficiency-based legislation can...
-
Suppose that 100 people live around a hazardous waste dump. If the people continue to live there for 20 years, one of them will likely contract a painful, nonfatal cancer that will lead to $1 million...
-
Surrounding the Great Lake are four paper mills, each producing 100 tons of paper per year. The paper is sold on the national market for $2 per ton, and including all the costs of production, costs...
-
In 2009, the conservative Heritage Foundation called the global-warming cap-andtrade bill being debated in the U.S. House a jobs destroyer and published an analysis claiming that the law, if passed,...
-
Explain the difference in determining the amount of income recognized from a conduit entity versus a taxable entity.
-
In the busy port of Chennai, India, the number of containers loaded onto ships during a 15-week period is as follows: 1. Develop a linear trend equation to forecast container loadings. 2. Using the...
-
Spermaceti, a fragrant substance from sperm whales, was much used in cosmetics until it was banned in 1976 to protect the whales from extinction. Chemically, spermaceti is cetyl palmitate, the ester...
-
The plasmalogens are a group of lipids found in nerve and muscle cells. How do plasmalogens differ fromfats? CH2OCH=CHR CR A plasmalogen H2OR"
-
What products would you obtain from hydrolysis of a plasmalogens (Problem 27.16) with aqueous NaOH with H3O+?
-
A New Life for Coca-Cola Excerpt and adaptation from an article published in The Globe and Mail, September 23 2016 The introduction of Coca-Cola Coke Life While Coca-Cola Co. has a number of newer...
-
The VP of Finance studied the printing costs at the university and knew it was time for bold, decisive action. The annual fixed cost of printing at the school was $850,000 and the per-page cost was...
-
What is the Herfindahl - Hirshman Index you calculate for this grocery industry? ( calculate to 4 decimal places ) Company Sales in $billion WALMART Inc ( includes Sam s Club ) $ 3 4 1 The Kroger Co...

Study smarter with the SolutionInn App


