State whether each of the following molecules is achiral or chiral. Cl H-C-Br -EL F (b) CI
Question:
State whether each of the following molecules is achiral or chiral.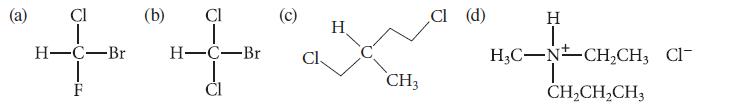
Transcribed Image Text:
Cl H-C-Br -EL F (b) CI H-C-Br Cl H CH3 Cl (d) H I H₂C-NCH₂CH, Cl- T CH₂CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a This compound is chiral b This compound is achiral c Thi...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Indicate whether each of the following statements is true or false. If false, explain why. (a) In some cases, constitutional isomers are chiral. (b) In every case, a pair of enantiomers have a mirror...
-
Indicate whether each of the following statements is true or false. If false, explain why. (a) In every case, pair of enantiomers have a mirror- image relationship. (b) If a compound has an...
-
Identify whether each of the following compounds is chiral or achiral: a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. CI
-
The 10-year Coupon Bond has a face value of $1,000, the annual coupon rate is 5 percent (out of its face value), the yield to maturity is 10 percent. (2.a) show me the cash flows of this coupon bond,...
-
Could a plaintiff win such a case without showing that she had contracted the HIV virus?
-
What are the typical characteristics of a company that should use a process costing system?
-
The codon change (Gly-12 to Val-12) in human H-ras that converts it to oncogenic H-ras has been associated with many types of cancers. For this reason, researchers would like to develop drugs to...
-
Impairment Assume the same information as E11-16, except that Pujols intends to dispose of the equipment in the coming year. It is expected that the cost of disposal will be $20,000. (a) Prepare the...
-
When estimating the average cash interest rate, Multiple choice question. the beginning amount of interest-bearing debt is in the denominator. the average amount of interest-bearing debt is in the...
-
Determine the stereochemical configuration of the following enantiomer of 3-chloro-1-pentene: HC CH Cl I CH CHCH3
-
Identify the asymmetric carbon(s) in 4-methyloctane: CH3 CH3CHCHCHCHCHCHCH3 4-methyloctane
-
Highlight the differences between ERP configuration, customization, and best of breed implementation strategies.
-
A Go Card account maintains a balance that may be spent on public transport. Users may request a statement that shows all transactions. The only transactions are to top up the account with some...
-
Finding zeros of a polynomial to d all real zeros of the function. h(x)=4(x+7)^(2)(x^(2)-9)(x-7)
-
What are the primary differences in accounting for for-profit businesses and nonprofit organizations? What some differences in accounting for governmental, nonprofit, and educational organizations?...
-
If you were to open your own business (a business that would require you to keep an inventory), what inventory costing method would you prefer (not FIFO)? Discuss why you chose the particular...
-
5 job descriptions for the accounting and payroll admin that you would apply for with a couple of sentences explaining why you chose them. Identify technical and specific skills as well as keywords...
-
What are the major determinants of price elasticity of demand? Use those determinants and your own reasoning in judging whether demand for each of the following products is probably elastic or...
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
Treatment of 1, 1-diphenyl- 1, 2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction. Ph H30+ PHCHCH Ph Ph
-
How would you prepare o-hydroxyphenyl-acetaldehyde from phenol? More than one step is required. HO o-Hydroxyphenylacetaldehyde CH-CO
-
Imagine that you have treated (2R, 3R)-2, 3-epoxy-3-methylpentane with aqueous acid to carry out a ring-opening reaction. (a) Draw the epoxide, showing stereochemistry. (b) Draw and name the product,...
-
Jenny's Novelties sells souvenir key chains at the local airport. JN sells these key chains at $12 each. The Variable Cost per chain is $8 and the Total Fixed Costs for teh year amounts to $13,000....
-
Roast House Coffee Importers sold 10 comma 000 units in October at a sales price of $ 5 per unit. The variable cost is $ 3 per unit. The monthly fixed costs are $ 15 comma 800. What is the operating...
-
National Foods has made its first ever issuance of 80,000 shares with a par value of $3 at $15 each. Its book value of net equity is $3,860,000. What is the retained earnings?

Study smarter with the SolutionInn App


