The following isomers do not differ greatly in stability. Predict which one should react more rapidly in
Question:
The following isomers do not differ greatly in stability. Predict which one should react more rapidly in an SN1 solvolysis reaction in aqueous acetone. Explain.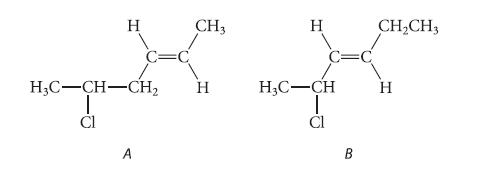
Transcribed Image Text:
H C=C A CH3 H3C-CH-CH₂ H Cl H H₂C-CH Cl B CH₂CH3 H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
According to Hammonds postulate Sec 48D of the text the rea...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) The following resonance-stabilized ion can protonate to give two different constitutional isomers. Give their structures and give the curved-arrow notation for their formation. (b) One of the...
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
Why was the value of China's currency a dominant issue at the recent U.S. - China summit? Why is the United States pushing for a higher renminbi? Why is China reluctant to allow its currency to...
-
A firm only uses one input in its production function, labor (L = number of workers) to produce car rides (all the workers provide their own cars, gas, etc.). The firm's total revenue (TR) function...
-
In what order are assets listed on the balance sheet?
-
Paul Adams owns a health club in downtown Los Angeles. He charges his customers an annual fee of $500 and has an existing customer base of 600. Paul plans to raise the annual fee by 6 percent every...
-
Taj Mahabub was the founder and CEO of GenAudio, a Colorado-based audio technology company. GenAudio had struggled financially practically since its formation, and Mahabub wished to secure a...
-
The trial balance of Nixon Company as of January 31, 2016, after the company completed the first month of operations, is shown in the partial worksheet below. INSTRUCTIONS 1. Record the trial balance...
-
Assumethe following attributes :A, B, C, D, E, F and Gwiththe candidate key :Aand Cand the following functional dependencies : A--> B, F A, C-->D, E F --> G a. Draw the 1NF relational model and...
-
Use a Frost circle to determine the -electron structure of (a) The cyclopentadienyl anion, which has a planar structure and six electrons; and (b) The cyclopropenyl cation, which has two electrons....
-
Suggest structures for the two constitutional isomers formed when 1,3-butadiene reacts with one equivalent of Br 2 . (Ignore any stereochemical issues.) Which of these products would predominate if...
-
What are the two main categories of functional budgets? Why do you think this might be a useful way to budget?
-
Why is being able to do arithmetic and algebra an essential skill for an accountant?
-
List some key differences in capital budgeting as applied to foreign versus domestic operations.
-
Discuss the relative importance of the final stage of the marketing research processcommunicating insightsin relation to the other stages.
-
On what two factors does the return on a foreign investment depend?
-
How do data visualization tools/dashboards and marketing research reports provide complementary knowledge?
-
Determine the amount of gross income Elbert must recognize in each of the following situations: a. In October, Elbert sells city of Norfolk bonds with a face value of $6,000 for $5,800. Elbert had...
-
(a) Prove that form an orthonormal basis for R3 for the usual dot product. (b) Find the coordinates of v = (1, 1, 1)T relative to this basis. (c) Verify formula (5.5) in this particular case. 48-65...
-
Propose structures for compounds that fit the following descriptions: (a) A hydrocarbon with seven lines in its 13C NMR spectrum (b) A six-carbon compound with only five lines in its 13C NMR spectrum...
-
Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure). TMS CH-cH 2 1 120 200 180 160 140 100 20 40 O ppm 60 Chemical shift (8) Intensity
-
Assign a chemical shift to each carbon in 6.methyl-5-hepten-2-ol(figure). (a) OH 0 ppm 200 180 160 140 120 100 80 60 40 20 Chemical shift (8) (b) 200 180 120 160 100 60 140 80 0 ppm Chemical shift...
-
How can a presentation of a team resume be drawn from the following factors? Team Resume: Security specialist User design expert Work closely with the banks? Email expert? Our team of software...
-
Carrie earns $11 per hour and works a 40 hour week. At the end of a week, she cashes her paycheck and then buys two tickets to a concert. If the concert tickets are $64 each, how much does she have...
-
Many lottery games for very large prizes include guessing which numbers will appear from a random sample of the numbered balls. In Minnesota's Gopher 5 lottery, there are five balls numbered 1-47. If...

Study smarter with the SolutionInn App


