Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure). TMS CH-cH 2 1 120
Question:
Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure).
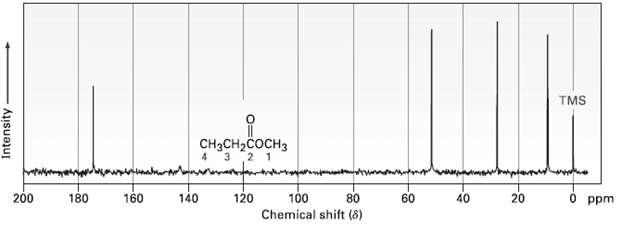
Transcribed Image Text:
TMS CнясH-cосHз 2 1 120 200 180 160 140 100 20 40 O ppm 60 Chemical shift (8) Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Methyl propanoate has 4 unique carbons and each one absorbs in a specific region of the ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The standard 13C NMR spectrum of phenyl propanoate is shown here. Predict the appearance of the DEPT-90 and DEPT-135 spectra. 13C NMR 0-C-CH2CH3 pheny! propanoate 200 180 160140 10 100 80 40 20 0 8...
-
The mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this hydrocarbon, and explain the spectraldata. 100 80 60 40 20 aly 120 10 40 60 80 100 140 m/z TMS 200 180...
-
3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 ?. Why are the two methyl groups attached to C3 nonequivalent? Making a molecular model should be...
-
The preferred stock of Walter Industries Inc. currently sells for $36 a share and pays $2.50 in dividends annually. What is the firms cost of capital for the preferred stock?
-
In most models of entry deterrence, the incumbent engages in predatory practices that harm a potential entrant. Can these models be reversed, so that the entrant engages in predatory practices? If so...
-
Based on their respective van der Waals constants (Table 10.3), is Ar or CO2 expected to behave more nearly like an ideal gas at high pressures? Explain. TABLE 10.3Van der Waals Constants for Gas...
-
What is the significance of industry comparables in valuation?
-
The trial balance of Pollard Company at the end of its fiscal year, August 31, 2012, includes these accounts: Beginning Inventory $18,700; Purchases $154,000; Sales Revenue $190,000; Freight-in...
-
What are three top nutritional considerations I should keep in mind when caring for a person with Autism?
-
You, CPA, work as a consultant on various engagements. Your client, Over The Edge Ltd. (OTE), has grown from a small custom snowboard manufacturer servicing the local market to a multinational...
-
Propose structures for compounds that fit the following descriptions: (a) A hydrocarbon with seven lines in its 13C NMR spectrum (b) A six-carbon compound with only five lines in its 13C NMR spectrum...
-
Assign a chemical shift to each carbon in 6.methyl-5-hepten-2-ol(figure). (a) OH 0 ppm 200 180 160 140 120 100 80 60 40 20 Chemical shift (8) (b) 200 180 120 160 100 60 140 80 0 ppm Chemical shift...
-
What is the purpose of the third standard of field work?
-
The price of a European call that expires in six months and has a strike price of $50 is $4. The underlying stock price is $52, and pays no dividends. The term structure is flat, with all risk-free...
-
On April 1 , 2 0 2 2 , ARGENTINA CORP. received a land from the government with a condition that a factory building should be constructed on it . On that date, the land has a fair value of P 6 , 0 0...
-
A trader creates a bull call spread by buying an option for $12.00 at the $100 strike price and selling an option at $5.00 at the $120 strike price. What is the net payoff per share (enter 4.00, not...
-
An investor has $5,000 invested in stock A and $5,000 in stock B. The daily volatilities of A and B are 0.5% and 1.50% respectively and the coefficient of correlation is 0.60. What is the one day 99%...
-
XYZ stock is trading at $700/share. A trader buys a long straddle in XYZ stock by buying a 700 strike call option for 60.00 and a 700 strike put option for 50.00, a total of $110. What are the...
-
For the vectors \(\vec{A}=2.0 \hat{\imath}+3.0 \hat{\jmath}\) and \(\vec{B}=-4.0 \hat{\imath}+5.0 \hat{\jmath}\), calculate \((a) \vec{A}+\vec{B}\) and (b) \(\vec{A}-\vec{B}\). (c) If the direction...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Use the vant Hoff factors in Table 14.9 to calculate each colligative property: a. The melting point of a 0.100 m iron(III) chloride solution b. The osmotic pressure of a 0.085 M potassium sulfate...
-
Predict the esterification products of the following acid/alcohol pairs. (a) CH3CH2CH2COOH + CH3OH (b) CH3OH + HNO3 (c) 2 CH3CH2OH + H3PO4 (d) (e) COOH + CH,CH,OH
-
Both cis- and trans-2-methylcyclohexanol undergo dehydration in warm sulfuric acid to give 1-methylcyclohexene as the major alkene product. These alcohols can also be converted to alkenes by...
-
Show how you would convert (S)-hexan-2-ol to (a) (S)-2-chlorohexane (b) (R)-2-bromohexane (c) (R)-hexan-2-ol?
-
A horizontal force of 5 newton is applied to a 2 kg block initially at rest on a rock horizontal surface. what is it's acceleration if the coefficient of friction is 0 . 2 ?
-
An equilateral triangle, as the drawing shows. The magnitude of each of the charges is 4 . 5 ???? ???? , and the lengths of the sides of the triangle are 1 . 5 ???? ???? . Calculate the magnitude of...
-
James and Esther Johnson are husband and wife and file a joint return. They live at 45678 S.W. 112th Street, Homestead, FL 33033. James, who is 67 years old (date of birth 12/14/1954), is retired and...

Study smarter with the SolutionInn App


