Using the curved-arrow notation, provide mechanisms for each of the reactions given in Fig. P22.91. (a) (b)
Question:
Using the curved-arrow notation, provide mechanisms for each of the reactions given in Fig. P22.91.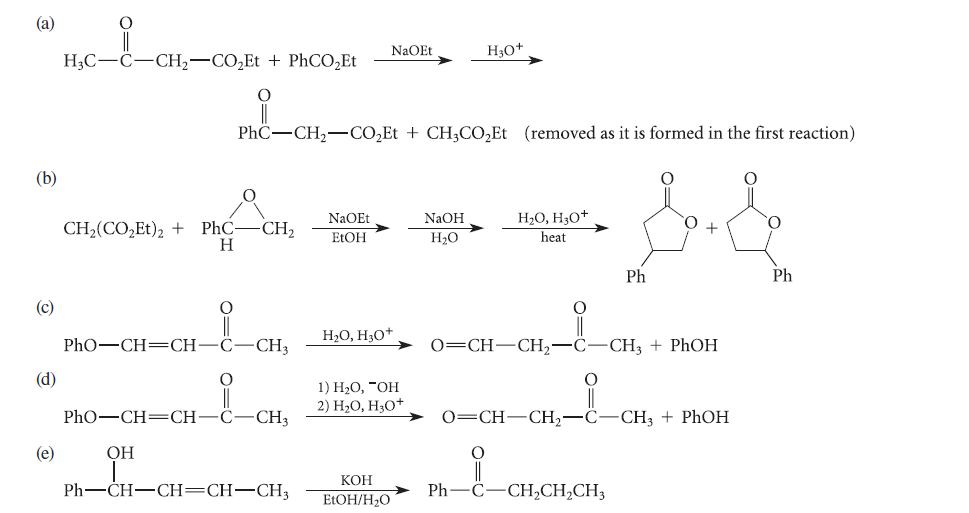
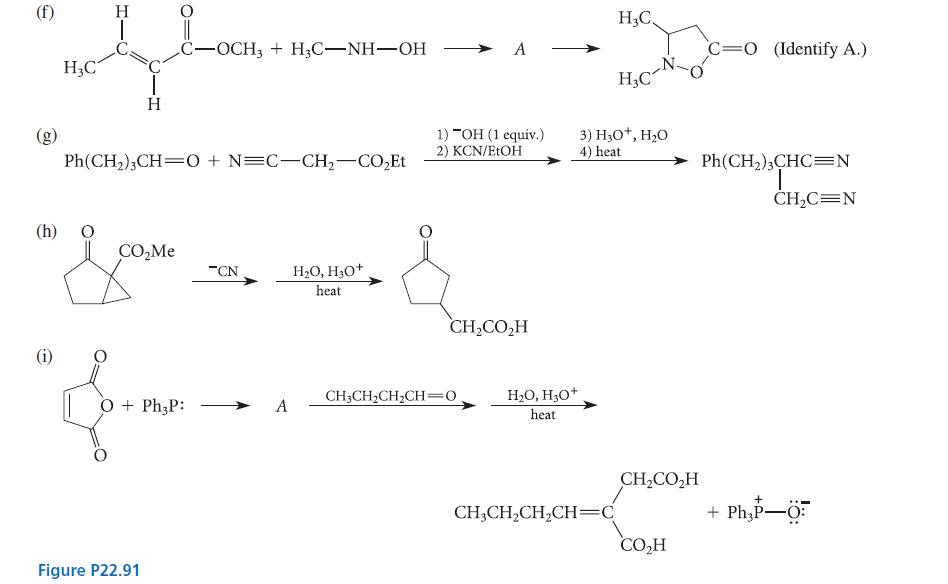
Transcribed Image Text:
(a) (b) (c) (d) (e) i H3C-C-CH₂-CO₂Et + PhCO₂Et CH,(CO,Et), + PhC- H PhO—CH=CH OH PhC-CH₂-CO₂Et + CH3CO₂Et (removed as it is formed in the first reaction) && PhO-CH=CH-C -CH3 CH₂ i -CH3 Ph—CH–CH=CH–CH NaOEt EtOH NaOEt H₂O, H₂O+ 1) H₂O, "OH 2) H₂O, H₂O+ KOH EtOH/H₂O H₂O+ NaOH H₂O H₂O, H₂O+ heat Ph O=CH-CH₂-C-CH3 + PhOH O=CH-CH₂-C-CH3 + PhOH i Ph—C–CH,CH,CH, Ph
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a A crossed Claisen condensation is followed by a reverse Claisen condensation driven by expulsion of the more volatile ester ethyl acetate from the reaction mixture The tertiary proton in compound A ...View the full answer

Answered By

Ashok Kumar Malhotra
Chartered Accountant - Accounting and Management Accounting for 15 years.
QuickBooks Online - Certified ProAdvisor (Advance - QuickBooks Online for 3 years.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the curved-arrow notation, provide mechanisms for each of the reactions given in Fig. P22.80 K-CO OH CH O (90% yield) CO CH CO,Et CH2a + 2 NaH - DMF NaOE CO.CH Dr C CO CIIs CO Et OH iri CO,Et...
-
Using the curved-arrow notation, provide mechanisms for each of the reactions given in Fig. P22.84. NaOEr Hio PhC-CHCO,Et +CH,CO,Et (removed as it is formed in the first reaction) NaOH Ph Ph CH3 CH3...
-
Using the curved-arrow notation, provide mechanisms for each of the reactions given in Fig. P22.87. H3C C-CH3 (c) (b) BrCHCO,CH3 I (CH)3 T BrCHCO,CH3 2 COEt KCO3 COEt NaOEt + 2 NaH HO CH3 DMF HO,...
-
How could sales force automation affect salesperson productivity, marketing management, and competitive advantage?
-
The accounts receivable turnover ratio is 8.14, and average net receivables during the period are $400,000.What is the amount of net credit sales for the period?
-
On January 1, Year 1, you are considering the purchase of Nico Enterprises' common stock . Based on your analysis of Nico Enterprises, you determine the following: 1. Book value at January 1, Year 1,...
-
Identify and describe the key attributes that might mitigate a red flag, suggesting that no bad act or compliance issues exist.
-
At January 1, 2010, Beidler Company reported retained earnings of $2,000,000. In 2010, Beidler discovered that 2009 depreciation expense was understated by $400,000. In 2010, net income was $900,000...
-
Consider that your cache is a direct mapped memory. It can hold up to 256 byte of data in 32-bit MIPS architecture. (a) Assuming that your block numbers, validity bits, and space allocated for tags...
-
A biochemist, Sal Monella, has come to you to ask your assistance in testing a promising biosynthetic hypothesis. She wishes to have two samples of methylsuccinic acid specifically labeled with 14 C...
-
When the epoxide 2-vinyloxirane reacts with lithium dibutylcuprate, followed by protonolysis, a compound A is the major product formed. Oxidation of A with PCC yields B, a compound that gives a...
-
Classic Auto Parts sells new and used auto parts. Although a majority of its sales are cash sales, it makes a significant amount of credit sales. During 2016, its first year of operations, Classic...
-
If the court decided to award Rebecca damages, what matters would the court take into consideration in assessing the amount she should be awarded?
-
What happens to the invention once a standard patent or an innovation patent has expired?
-
Embezzlement is regarded as a white-collar crime. Is it helpful to distinguish white-collar crimes from other types of criminal offenses? If so, why? Should white-collar criminals like Bernie Madoff...
-
The Malaysian government has recently moved to repeal the exemption granted to foreign-source income in stages. Critically debate about this move.
-
When discussing federal, state, and local agencies and their role: How can I explain the concepts of due process and the equal protection clause? What example can I provide of an administrative...
-
All-Canadian, Ltd. is a multiproduct company with three divisions: Pacific Division, Plains Division, and Atlantic Division. The company has two sources of long-term capital: debt and equity. The...
-
A line l passes through the points with coordinates (0, 5) and (6, 7). a. Find the gradient of the line. b. Find an equation of the line in the form ax + by + c = 0.
-
Specify the alkene and reagents needed to synthesize each of the following diols. (a) (b) (Recemic) (c) (Recemic) OH OH OH OH
-
Predict the products of the following ozonolysis reactions. (a) (b) (c) (1) O2 (2) Me2S (1) 03 (2) Me S (1) O3 (2) Me S
-
Write the structures of the alkenes that would yield the following carbonyl compounds When treated with ozone and then with dimethyl sulfide. (a) (b) (2 Mol is produced from 1 mol of alkene) (c) o...
-
Canadian honey producer Bee Maid Honey has been in business since 1954. They are a Western Canadian beekeeping cooperative that produces premium honey and is owned by more than 300 beekeepers. Liquid...
-
An industrial chemical plant produces hazardous by-products. The own or private marginal cost function (supply function) of this chemical producer can be represented as PMC = 5,000 + 0.25Q. Given...
-
: Apply risk management practices. Read the scenario and complete the tasks that follow. Scenario 3: Applying risk management practices. You are the branch manager at Hudson's Real Estate, Napier....

Study smarter with the SolutionInn App


