Using the data in Table 8.1, estimate the carbonselenium bond length in H 3 CSeCH 3 .
Question:
Using the data in Table 8.1, estimate the carbon–selenium bond length in H3C—Se—CH3.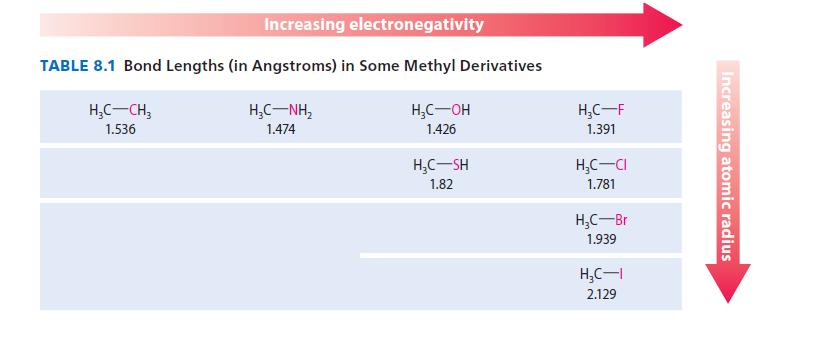
Transcribed Image Text:
Increasing electronegativity TABLE 8.1 Bond Lengths (in Angstroms) in Some Methyl Derivatives H₂C-CH₂ 1.536 HỌC—NH, 1.474 H₂C-OH 1.426 H₂C-SH 1.82 H₂C-F 1.391 H₂C-CI 1.781 H₂C-Br 1.939 H₂C-I 2.129 Increasing atomic radius
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
In Table 81 the bond lengths in a given period are about 004 greater in the ...View the full answer

Answered By

Robert Mbae
I have been a professional custom essay writer for the last three years. Over that period of time, I have come to learn the value of focusing on the needs of the clients above everything else. With this knowledge, I have worked hard to become an acclaimed writer that can be trusted by the customers to handle the most important custom essays. I have the necessary educational background to handle projects up to the Ph.D. level. Among the types of projects that I've done, I can handle everything within Dissertations, Project Proposals, Research Papers, Term Papers, Essays, Annotated Bibliographies, and Literature Reviews, among others.
Concerning academic integrity, I assure you that you will receive my full and undivided attention through to the completion of every essay writing task. Additionally, I am able and willing to produce 100% custom writings with a guarantee of 0% plagiarism. With my substantial experience, I am conversant with all citation styles ranging from APA, MLA, Harvard, Chicago-Turabian, and their corresponding formatting. With all this in mind, I take it as my obligation to read and understand your instructions, which reflect on the quality of work that I deliver. In my paper writing services, I give value to every single essay order. Besides, whenever I agree to do your order, it means that I have read and reread your instructions and ensured that I have understood and interpreted them accordingly.
Communication is an essential part of a healthy working relationship. Therefore, I ensure that I provide the client with drafts way long before the deadline so that the customer can review the paper and comment. Upon completion of the paper writing service, the client has the time and right to review it and request any adjustments before releasing the payment.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The Tastee Bakery Company supplies a bakery product to many supermarkets in a metropolitan area. The company wishes to study the effect of the height of the shelf display employed by the supermarkets...
-
Carbon disulfide (CS2) is a toxic, highly flammable substance. The following thermodynamic data are available for CS2(l) and CS2(g) at 298 K: (a) Draw the Lewis structure of the molecule. What do you...
-
Using the data in Table 4-3, calculate the G° for ring flip to the other conformation of the molecules depicted in Problem 30. Make sure that the sign (i.e., positive or negative) of your values...
-
In an organization, managers communicate information downward to their departments and teams, and employees communicate information upward to their managers. If all members of an organization are not...
-
TRUE/FALSE 1. A problem for many international merchants is that tariff s have been rising for the last decade. 2. The United States imports more goods and services (combined) than it exports. 3....
-
What is the statute of limitations, and what role does it play in the filing of tax returns?
-
Redesign the fractionator of Example 6.8 for a reflux ratio that is twice the minimum. Determine the diameter of the tower, the height of packing in the stripping and rectifying sections, and the...
-
At Dot Corn, a large retailer of popular books demand is constant at 32.000 books per year. The cost of placing an order to replenish stock is $10 and the annual cost of holding is $4 per book. Stock...
-
Based on the Lease Obligations by Year visualization, do the results make sense? Select answer from the options below No. There is a decreasing trend in finance lease and operating lease obligations,...
-
(a) A chemist used the name 3-butyl-1,4-dioxane in a paper. Although the name unambiguously describes a structure, what should the name have been? Explain. (b) Give the structure of 2-butoxyethanol,...
-
Draw the structure of each of the following compounds. (a) Ethyl propyl ether (b) Dicyclohexyl ether (c) Tert-butyl isopropyl sulfide (d) Allyl benzyl ether (e) Phenyl vinyl ether (f)...
-
Sketch the region enclosed by the given curves. Decide whether to integrate with respect to x or y. Draw a typical approximating rectangle and label its height and width. Then find the area of the...
-
A hedge portfolio consists of a dividendpaying stock and a forward contract to eliminate the uncertainty of the selling price of the stock. The forward price should be such that it forces the hedge...
-
Cruiseliners , Inc. has a beta of 1.5 and a cost of equity of 13.2 percent . The risk - free rate of return is 4.2 percent . Cruiseliners is considering a project with a beta of 1.7 and a project...
-
what is the impact on real estate agency if not banking client money into a trust account
-
Review shopifys, expansion activities and business growth of past ten years (approximately) and discuss the following 1. Do you think their selection of international locations/countries was...
-
A father has brought home two equal-sized doughnuts for his children Doug and Isabel. Doug says he loves doughnuts more than Isabel, who agrees, but says she likes them too. The father agrees with...
-
Suppose a firm expects that a $20 million expenditure on R&D will result in a new product that will increase its revenue by a total of $30 million 1 year from now. The firm estimates that the...
-
Sandcastles, Inc.s management has recently been looking at a proposal to purchase a new brick molding machine. With the new machine, the company would not have to buy bricks. The estimated useful...
-
Explain why neopentane shows no molecular ion is its mass spectrum, Predict the structure and m/z for these base ion in its massspectrum. CH3 CH .-, -C- CH3 Neopentane
-
Explain how the peaks at m/z 115, 101, and 73 arise in the mass spectrum of 3-methy-3-heptanol.
-
Suggest a structure for the compound whose mass spectrum is asfollows: 100 107 81 93 188 70 80 110 120 90 100 130 150 160 170 140 180 190 200 Relative abundance
-
A child, hunting for his favorite wooden horse, is running on the ground around the edge of a stationary merry-go-round. The angular speed of the child has a constant value of 0.222 rad/s. At the...
-
Bob says that (x + y) 2 = x^(2) + y^(2) is a shortcut rule of multiplication while Joe says that it doesn't work. Show who is correct by giving at least two different explanations (numeric,...
-
The penny-farthing is a bicycle that was popular between 1870 and 1890. As the figure shows, this type of bicycle has a large front wheel and a small rear wheel. On a Sunday ride in the park the...

Study smarter with the SolutionInn App


