When each of the compounds shown in Fig. P28.43 is heated in the presence of maleic anhydride,
Question:
When each of the compounds shown in Fig. P28.43 is heated in the presence of maleic anhydride, an intermediate is trapped as a Diels–Alder adduct. What is the intermediate formed in each reaction, and how is it formed from the starting material?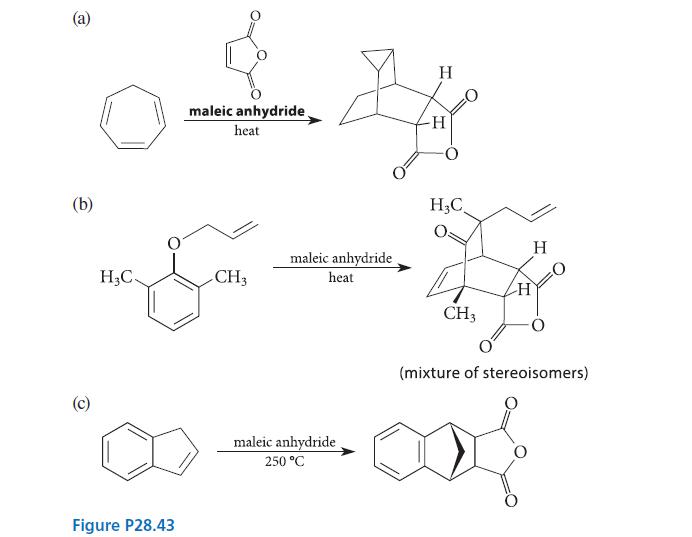
Transcribed Image Text:
(a) (b) (c) H₂C. maleic anhydride heat Son CH3 Figure P28.43 maleic anhydride heat maleic anhydride. 250 °C H H H₂C CH3 H H (mixture of stereoisomers)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a The approach to solving this type of problem is described in the solution to Pro...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many 13C NMR signals would you predict for each of the compounds shown in Problem 9.22?
-
What compounds would you expect to be formed when each of the following ethers is refluxed with excess concentrated hydrobromic acid? (a) (b) (c) (d) (THF) (1,4-dioxane)
-
What product(s) are expected when each of the following compounds reacts with one equivalent of NBS in CC14 in the presence of light and peroxides? Explain your answers. (a) cyclohexene (b)...
-
Nanette works for Piroz and is paid a basic wage of $1,000 a week. Piroz operates the following bonus scheme: (1) Each employee gets a bonus of $4 for every unit they produce in excess of 2,000 units...
-
You are considering investing the cash gifts you received for graduation in various stocks. You have received several annual reports of major companies. Required: For each of the following, indicate...
-
Martin Manufacturing manufactures ready- mix cement with the following standard costs: Direct materials ................................................... 2.5 pounds at $ 2.40 per pound Direct labor...
-
Petitioner Christy Brzonkala met respondents Antonio Morrison and James Crawford at a campus party at Virginia Polytechnic Institute (Virginia Tech), where they were all students. At the party, the...
-
Holiday Fruit Company buys oranges and processes them into gift fruit baskets and fresh juice. The company grades the fruit it buys on a scale from 1 (lowest quality) to 5 (highest quality). The...
-
8. Two dice are rolled. Let A represent rolling a sum greater than 7. Let B represent rolling a sum that is a multiple of 3. Determine n(AB). A. 5 B. 8 C. 12 D. 15 Long Answer: Show all work for the...
-
A compound A (C 11 H 14 O 3 ) is insoluble in base and gives an isomeric compound B when heated strongly. Compound B gives a sodium salt when treated with NaOH. Treatment of the sodium salt of B with...
-
Certain black bugs of the order Hemiptera, generally observed in the tropical regions of India immediately after the rainy season, give off a characteristic nauseating smell whenever they are...
-
Deduce the virial expansion (7.1.13) from equations (7.1.7) and (7.1.8), and verify the quoted values of the virial coefficients.
-
Suppose the address of the first instruction is 245101AO in hexadecimal in the following code. Show in decimal the immediate value (i.e. offset) in the beq and bne instruction, and in hexadecimal (of...
-
- Write a program that takes in two matrixes - A and B. A user will enter the matrixes. Both matrixes are 3X3 Multiply matrix A by B. Use an multi-dimensional "array" Display the output in a matrix...
-
A refrigerator operating reversibly extracts 45.0 kJ of heat from a thermal reservoir and delivers 67.0 kJ as heat to a reservoir at 300 K. Calculate the temperature of the reservoir from which heat...
-
Glider A , traveling at 8 . 4 m / s on an air track, collides perfectly elastically with glider B traveling at 4 . 7 m / s in the same direction. The gliders are of equal mass. What is the final...
-
A circuit contains logic between two registers and is intended to run at 500MHz. The parameters for the circuit are: TLogic = 300 to 800ps TCIK-Q = 400 to 500ps TSetup = 300ps T Hold = 50ps Calculate...
-
Should a retail firm include the cost of receiving and stocking goods when computing inventory values?
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
The nucleosides shown in Figs. 25.4 and 25.5 are stable in dilute base. In dilute acid, however, they undergo rapid hydrolysis yielding a sugar (deoxyribose or ribose) and a heterocyclic base. (a)...
-
Basing your answer on reactions that you have seen before, propose a likely mechanism for the condensation reaction in the first step of the preceding uridine synthesis.
-
(a) What kind of linkage is involved in the acetonide group of the protected nucleoside, and why is it susceptible to mild acid-catalyzed hydrolysis? (b) How might such a protecting group be...
-
Naturally, the manager and staff did not receive a bonus. However, the manager was upset with this turn of events. "We all worked extra hard this year. It was a tough year. The fuel prices more than...
-
Green Caterpillar's accumulated owed financial obligations decreased from Year 1 to Year 2. This statement is , because: Accruals actually increased from $0 in Year 1 to $410 million at the end of...
-
To provide incentives, the NWC made the supervisors in Montreal and at the winter trading posts partners. A negative aspect of this organizational form was Group of answer choices the NWC was cash...

Study smarter with the SolutionInn App


