When S-ethyl thiobenzoate (A) is allowed to react with 2-aminoethanol (B), an amide C is formed if
Question:
When S-ethyl thiobenzoate (A) is allowed to react with 2-aminoethanol (B), an amide C is formed if one equivalent of triethylamine (Et3N:) is included in the reaction mixture, but an ester D is formed if one equivalent of a strong acid such as p-toluenesulfonic acid is added as a catalyst. (See Fig. P25.25.) Give the structures of C and D, and explain why different products are formed under different conditions.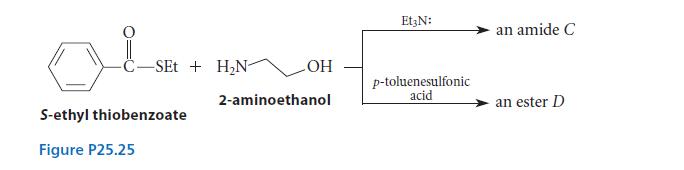
Transcribed Image Text:
C-SEt + H₂N- S-ethyl thiobenzoate Figure P25.25 OH 2-aminoethanol Et3N: p-toluenesulfonic acid an amide C an ester D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
When base triethylamine is present the product is an amide because the amine end of 2aminoe...View the full answer

Answered By

Muhammad Ahtsham Shabbir
I am a professional freelance writer with more than 7 years’ experience in academic writing. I have a Bachelor`s Degree in Commerce and Master's Degree in Computer Science. I can provide my services in various subjects.
I have professional excellent skills in Microsoft ® Office packages such as Microsoft ® Word, Microsoft ® Excel, and Microsoft ® PowerPoint. Moreover, I have excellent research skills and outstanding analytical and critical thinking skills; a combination that I apply in every paper I handle.
I am conversant with the various citation styles, among them; APA, MLA, Chicago, Havard, and AMA. I also strive to deliver the best to my clients and in a timely manner.My work is always 100% original. I honestly understand the concern of plagiarism and its consequences. As such, I ensure that I check the assignment for any plagiarism before submission.
4.80+
392+ Reviews
587+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
When methyl iodide at 0.I M concentration is allowed to react with sodium ethoxide at 0.1 M concentration in ethanol solution, the product ethyl methyl ether is obtained in good yield. Explain why...
-
The following "cycle of copper" experiment is performed in some general chemistry laboratories. The series of reactions starts with copper and ends with metallic copper. The steps are as follows: (1)...
-
On 1 June 1998, Alice bought a house in Derby for 45,000. - She occupied the house as her PPR until 1 May 2000 when she left to work in Exeter, living in rented accommodation. - She returned to the...
-
The isotope 14 C decays according to 14 C 14 N + e + v e . The atomic mass of 14 N is 14.003074 u. Determine the maximum kinetic energy of the electron.
-
A firm with a 13% cost of capital must select the optimal group of projects from those shown in the following table, given its capital budget of $1 million. a. Calculate the present value of cash...
-
Discuss how perceptions of organizational support can influence employees attitudes and behaviour. What can organizations do to develop positive perceptions of organizational support?
-
1. Go back to the ethical dilemma at the beginning of the case. Which position did you take and why? Did your position change after you read the case? 2. Was Chiquita justified in making the...
-
i. Write the program in C++ Write a four-function calculator to perform addition, subtraction, multiplication & division that takes two operands and an operator from user. Perform this logic by using...
-
When S-cyclohexyl thioacetate is reduced by LiAlH 4 in ether, followed by protonolysis, cyclohexanethiol is formed. However, when a large excess of the Lewis acid BF 3 is added to the reaction...
-
Complete the reactions shown in Fig. P25.24 by drawing the structures of the products, and explain your reasoning. (b) (c) afa CI H3C- + HN(CH,), (large excess) Holofo Figure P25.24 (1) TsCl OH...
-
Derive the form of the excess enthalpy predicted by Wilsons equation assuming that A ij s and ratios of molar volumes are temperature-independent.
-
Consider the history of the world from 1 million b.c. until now. Although limited data exists from early history, we still have some information about how people lived and how much they consumed. a....
-
The cost of the average consumers basket of goods and services in 2022 is roughly 12 times what it was in 1950. In other words, what the average consumer bought for $100 in 1950 would cost a consumer...
-
Sophia operates her own accounting practice and is looking to hire two entrylevel accountants. A high-productivity worker will generate $90,000 in revenue per year and a low-productivity worker will...
-
Real GDP per person falls during a devastating recession, which in turn causes consumption to fall by 5%. Would giving everyone 5% more income during the recession lead consumption to go up 5%? Why...
-
When you rent an apartment or house, most landlords will require that you pay a refundable security deposit. Why do you think landlords do this? Relate their actions to possible problems caused by...
-
A survey of 2,645 consumers by DDB Needham Worldwide of Chicago for public relations agency Porter/Novelli showed that how a company handles a crisis when at fault is one of the top influences in...
-
The tractor is used to lift the 150-kg load B with the 24-mlong rope, boom, and pulley system. If the tractor travels to the right at a constant speed of 4 m/s, determine the tension in the rope when...
-
Give the structure of the only product formed when diethyl -methyladipate (compound C) reacts in the Dieckmann condensation. Explain your reasoning. Eto,C( CH2) 2Et CH3
-
Complete the following reaction. Assume that one equivalent of NaOEt is present in following case. Ph-C-CH3 + Ph-C-OEt (excess) 0,
-
Analyze each of the following compounds and determine what starting materials would be required for its synthesis by a Claisen condensation. Then decide which if any of the possible Claisen...
-
11: Let's calculate the average useful power output for the following three situations: (a) A 75 kilogram sprinter accelerates from zero to 11 meters per second in a time of 3.0 seconds (b) A 75...
-
8. A solid cylinder with a mass of m and radius of R is rolling down an inclined plane without slipping from the height of h. The gravitational acceleration is g. Find the translational velocity (v)...
-
A newly manufactured product has a total cost of $11,000,000. They produced 2,000,000 units that cost on average $2.50 each to make. How much is the company's fixed costs?

Study smarter with the SolutionInn App


